使用顺序:先背 Frequency 高的行,再用 Sources 回到原题核对题干。统计范围为 2020-2025 AS Paper 2 mark schemes,N = 44;同一卷同一点只计 1 次。Full-mark wording 是可直接背诵的答案骨架;Sources 已按最新到最旧排列,MS 文件名通常把
_qp_换成_ms_。no 2020-2025 P2 MS hit表示这条是 syllabus / core review point,放在 appendix,不参与高频排序。
考试定位:CIE 9701 AS Chemistry Paper 2 是 AS Level Structured Questions 理论卷;考试时间 1 h 15 min,60 marks。题目覆盖 AS syllabus 的 physical / inorganic / organic chemistry,常见题型是 definitions, explanations, calculations, equations, data interpretation 和 organic mechanisms。
Official syllabus: Cambridge 9701 Chemistry syllabus 2025-2027; Paper 2 is a written paper, 1 hour 15 minutes, 60 marks.
答题核心:按 MS 关键词拿分。定义题写必要限定词;解释题按原因 → 作用对象 → 结果写完整因果链;计算题保留单位和合适 s.f.;有机机理题要画清 curly arrows 的起点、终点、电荷和中间体。
Highest-Frequency First
| Frequency | Papers | Chapter | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 64% | 28/44 | 14-17 | mechanism curly-arrow rule | 1. curly arrows start at a lone pair/electron pair/bond/negative charge 2. arrow points to the atom or bond receiving the electron pair 3. for bond breaking, arrow goes from the bond to the atom receiving the electrons |
9701_w25_qp_24 Q3(b)(ii); 9701_s25_qp_22 Q3(a)(ii); 9701_m25_qp_22 Q4(b)(i); +25 additional hits |
| 59% | 26/44 | 3 | bonding diagrams: metallic / dot-and-cross / coordinate | 1. for dot-and-cross diagrams, show outer-shell electrons only and use different symbols for electrons from different atoms 2. covalent bonds are shared pairs; coordinate/dative bonds have both electrons in the shared pair donated by one atom/species 3. metallic bonding diagrams need a lattice/regular arrangement of positive ions/cations and delocalised electrons 4. for NH₄Cl, show ionic attraction between NH₄⁺ and Cl⁻, four N-H covalent bonds in NH₄⁺, and one N→H coordinate bond / one N-H bonding pair from N |
9701_s24_qp_23 Q4(a); 9701_w20_qp_21 Q3(a)(i); 9701_s20_qp_22 Q1(b); +23 additional hits |
| 50% | 22/44 | 3 | simple molecular boiling/melting | 1. greater number of electrons in the molecules 2. stronger instantaneous dipole-induced dipole forces / London dispersion forces / van der Waals’ forces between molecules 3. instantaneous dipoles induce dipoles in neighbouring molecules, producing attraction between δ⁺ and δ⁻ ends 4. more energy is needed to overcome the stronger intermolecular forces |
9701_w20_qp_23 Q4(a)(iii); 9701_w20_qp_21 Q4(a)(iii); 9701_s20_qp_21 Q1(b)(i); +21 additional hits |
| 43% | 19/44 | 3 / 13 | σ and π bonds / orbital overlap / hybridisation | 1. a σ bond is formed by direct/head-on/end-on overlap of orbitals between the bonding atoms 2. a π bond is formed by sideways/lateral overlap of adjacent p orbitals 3. single bond = 1σ; double bond = 1σ + 1π; triple bond = 1σ + 2π 4. common hybridisation: sp gives linear/triple-bond centres; sp² gives trigonal planar/C=C centres; sp³ gives tetrahedral/saturated carbon or ammonium centres |
9701_s24_qp_23 Q1(a)(ii); 9701_s24_qp_21 Q4(b)(i-ii); 9701_s22_qp_21 Q2(a); +16 additional hits |
| 41% | 18/44 | 21 | oxidation/reduction route | 1. choose reagent and conditions from the functional group 2. acidified K₂Cr₂O₇/KMnO₄ with distil/reflux for alcohols/aldehydes 3. NaBH₄ or LiAlH₄ for carbonyl reduction |
9701_w20_qp_21 Q3(c)(i); 9701_s20_qp_22 Q5(a); 9701_m20_qp_22 Q3(a)(i); +22 additional hits |
| 36% | 16/44 | all | equations / state symbols | 1. use correct formulae 2. balance the equation 3. include charges where needed 4. include state symbols when the question asks for them |
9701_s20_qp_22 Q1(e)(ii); 9701_s20_qp_21 Q2(a)(i); 9701_s20_qp_21 Q1(c)(i); +20 additional hits |
| 36% | 16/44 | 1 | IE trend explanation | 1. compare nuclear charge/proton number 2. compare distance/atomic radius and shielding by inner shells or sub-shells 3. stronger attraction for the outer electron gives a higher ionisation energy 4. exceptions: Al is lower than Mg because its electron is removed from 3p rather than 3s; S is lower than P because paired 3p electrons repel each other (spin-pair repulsion) |
9701_w20_qp_22 Q1(b)(iv); 9701_w20_qp_21 Q1(c); 9701_w20_qp_21 Q1(b); +24 additional hits |
| 36% | 16/44 | 22 | IR absorption identification | 1. quote the absorption range in cm⁻¹ 2. state the bond responsible 3. state the functional group responsible 4. use presence/absence of O-H, C=O, C≡N etc. as evidence |
9701_w20_qp_21 Q3(c)(v); 9701_s20_qp_21 Q6(e); 9701_m20_qp_22 Q3(d)(iii); +17 additional hits |
| 32% | 14/44 | 9 | Period 3 melting point structure | 1. identify the structure: simple molecular / giant metallic / giant covalent as appropriate 2. simple molecular substances: only weak intermolecular / van der Waals’ forces are overcome, so less energy is needed 3. giant metallic/covalent substances: many strong metallic/covalent bonds or attractions must be overcome/broken, so more energy is needed |
9701_w21_qp_23 Q3(a)(i); 9701_w21_qp_21 Q3(a)(i); 9701_w20_qp_22 Q2(f); +12 additional hits |
| 32% | 14/44 | 6 | reducing agent | 1. donates electrons and is oxidised | 9701_w20_qp_22 Q4(b)(iii); 9701_w20_qp_21 Q3(c)(ii); 9701_m20_qp_22 Q2(c)(ii); +14 additional hits |
| 32% | 14/44 | 3 | metallic bonding / conductivity | 1. giant metallic lattice / regular arrangement of positive ions/cations surrounded by delocalised electrons 2. many strong electrostatic attractions between cations and delocalised electrons / strong metallic bonds need more energy to overcome, so melting point is high 3. delocalised electrons are free to move through the structure and carry charge |
9701_w24_qp_21 Q3(a)(iii); 9701_s22_qp_23 Q1(f)(ii); 9701_s22_qp_22 Q1(a); +5 additional hits |
| 30% | 13/44 | 11 | chlorine disproportionation / water purification | 1. cold dilute NaOH: Cl₂ + 2NaOH → NaCl + NaClO + H₂O 2. hot concentrated NaOH: 3Cl₂ + 6NaOH → 5NaCl + NaClO₃ + 3H₂O 3. in water: Cl₂ + H₂O ⇌ HCl + HOCl 4. HOCl kills bacteria/microbes in water purification; ClO⁻ is the active species in alkaline bleach 5. chlorine disproportionates because Cl is both oxidised and reduced |
9701_m25_qp_22 Q3(b); 9701_s23_qp_22 Q2(d); 9701_s21_qp_23 Q1(c); +10 additional hits |
| 27% | 12/44 | 14 | major product / carbocation stability | 1. major product forms via the more stable carbocation/intermediate 2. more alkyl groups give greater positive inductive effect/electron donation 3. positive charge is spread/stabilised more |
9701_s20_qp_21 Q5(d)(ii); 9701_s20_qp_21 Q5(d)(i); 9701_m20_qp_22 Q3(c)(iii); +30 additional hits |
| 27% | 12/44 | 7 | Kp / Kc expression and units | 1. for aA + bB ⇌ cC + dD, write Kc = [C]c[D]d / ([A]a[B]b) 2. for gases, write Kp = p(C)cp(D)d / (p(A)ap(B)b) 3. products over reactants; powers are stoichiometric coefficients 4. omit pure solids/liquids; derive units from the final expression |
9701_s23_qp_23 Q4(b)(ii); 9701_s21_qp_23 Q2(c)(iii); 9701_m20_qp_22 Q1(f)(ii); +9 additional hits |
| 27% | 12/44 | 3 | bond polarity / permanent dipole forces | 1. a polar bond forms when bonded atoms have different electronegativities 2. the more electronegative atom attracts the bonding pair more strongly and becomes δ⁻; the other atom becomes δ⁺ 3. permanent dipole-permanent dipole forces act between polar molecules when δ⁺ and δ⁻ ends attract |
9701_w25_qp_23; 9701_w25_qp_21; 9701_s25_qp_22; 9701_m25_qp_22; +8 additional hits |
| 25% | 11/44 | 12 | SO₂ acid rain | 1. SO₂ reacts with water to form H₂SO₃ 2. SO₂ / H₂SO₃ can be oxidised to H₂SO₄ 3. these acids cause acid rain |
9701_w21_qp_23 Q1(d)(ii); 9701_w21_qp_21 Q1(d)(ii); 9701_s20_qp_22 Q2(d); +3 additional hits |
| 25% | 11/44 | 6 | oxidising agent | 1. accepts electrons and is reduced | 9701_m21_qp_22 Q4(c)(i); 9701_m20_qp_22 Q2(c)(ii); 9701_m20_qp_22 Q2(b)(i); +11 additional hits |
| 25% | 11/44 | 7 | Bronsted acid / strong vs weak acid | 1. acid = proton/H⁺ donor 2. strong acid fully dissociates/fully ionises in aqueous solution/water 3. weak acid partially dissociates/partially ionises in aqueous solution/water |
9701_w20_qp_23 Q2(b)(i); 9701_w20_qp_22 Q3(d)(ii); 9701_w20_qp_21 Q2(b)(i); +11 additional hits |
| 23% | 10/44 | 14 | alkene test | 1. bromine water / Br₂(aq) is decolourised from orange/brown to colourless | 9701_s22_qp_22 Q6(a); 9701_s21_qp_21 Q5(c)(ii); 9701_s21_qp_21 Q4(b); +7 additional hits |
| 23% | 10/44 | 17 | 2,4-DNPH test | 1. 2,4-DNPH gives a red/orange/yellow precipitate with carbonyl compounds (aldehydes/ketones) | 9701_s21_qp_23 Q4(b); 9701_w20_qp_22 Q4(c); 9701_s20_qp_21 Q6(d); +10 additional hits |
| 23% | 10/44 | 19 | nitrile hydrolysis to carboxylic acid / carboxylate | 1. acid hydrolysis: RCN + 2H₂O + HCl/H⁺ → RCOOH + NH₄Cl/NH₄⁺ 2. alkaline hydrolysis: RCN + 2H₂O + NaOH → RCO₂Na + NH₄OH/NH₃; acidification gives RCOOH if required 3. heat/reflux with dilute acid or aqueous alkali 4. the nitrile carbon becomes the carboxyl carbon, so the carbon chain length does not change during hydrolysis |
9701_s25_qp_24 Q4(d)(i); 9701_s22_qp_22 Q5(c)(ii); 9701_w20_qp_21 Q3(c)(iv); +7 additional hits |
| 20% | 9/44 | 8 | frequency of effective collisions | 1. rate increases/decreases with the frequency of successful/effective collisions per unit time | 9701_s22_qp_21 Q1(g); 9701_m22_qp_22 Q2(b)(iii); 9701_m21_qp_22 Q1(c)(ii); +7 additional hits |
| 20% | 9/44 | 11 | NOₓ / atmospheric pollution | 1. NOₓ forms when N₂ and O₂ react at high temperature in an internal combustion engine / lightning 2. NO/NO₂ reacts with oxygen and/or water/moist air to form HNO₂/HNO₃ and cause acid rain 3. NO/NO₂ can act as a homogeneous catalyst in SO₂ oxidation to SO₃ and is regenerated 4. NO₂/NOₓ reacts with unburned hydrocarbons/VOCs to form photochemical smog / PAN |
9701_s24_qp_23 Q2(d)(i-ii); 9701_s23_qp_21 Q5(b)(i-iii); 9701_s22_qp_21 Q2(b)(i-ii); 9701_s20_qp_22 Q2(d); +5 additional hits |
| 20% | 9/44 | 9 | Period 3 chloride hydrolysis | 1. SiCl₄ + 2H₂O → SiO₂ + 4HCl 2. PCl₅ + 4H₂O → H₃PO₄ + 5HCl 3. include state symbols / acidic products if the question asks |
9701_w21_qp_23 Q3(b)(ii); 9701_w21_qp_21 Q3(b)(ii); 9701_w20_qp_22 Q3(b)(ii); +12 additional hits |
| 20% | 9/44 | 17 | Tollens / Fehling distinction | 1. aldehydes reduce Tollens’ reagent to a silver mirror/grey precipitate 2. aldehydes reduce Fehling’s solution to a brick-red precipitate 3. ketones show no visible change |
9701_m22_qp_22 Q4(a)(i); 9701_m21_qp_22 Q4(b); 9701_w20_qp_22 Q4(a)(iv); +6 additional hits |
| 20% | 9/44 | 2 | relative atomic/isotopic/molecular mass | 1. relative isotopic mass = mass of one atom of an isotope compared with 1/12 of the mass of a carbon-12 atom 2. relative atomic mass = weighted mean mass of atoms of an element compared with 1/12 of carbon-12 3. relative molecular/formula mass = sum of the relative atomic masses in the molecule/formula unit |
9701_w22_qp_21 Q1(a)(ii); 9701_s20_qp_23 Q2(a); 9701_s20_qp_22 Q1(a); +6 additional hits |
| 20% | 9/44 | 1 | ionic radius / isoelectronic comparison | 1. compare occupied electron shells first 2. in an isoelectronic series, ions have the same number of electrons / same electron arrangement 3. the ion with more protons has a smaller ionic radius because the same electron cloud is attracted more strongly 4. down a group, ionic radius increases because there are more shells and more shielding |
9701_w25_qp_23; 9701_w25_qp_21; 9701_s25_qp_24; 9701_s25_qp_23; +5 additional hits |
| 18% | 8/44 | 14 | free-radical substitution | 1. initiation: homolytic fission in UV light forms halogen radicals 2. propagation 1: a halogen radical removes H from an alkane to form an alkyl radical and HCl/HBr 3. propagation 2: the alkyl radical reacts with Cl2/Br2 to form a halogenoalkane and regenerate a halogen radical 4. termination: two radicals combine to form a stable molecule |
9701_w24_qp_21 Q2(b)(iii); 9701_m21_qp_22 Q2(g)(i); 9701_s20_qp_23 Q4(b)(ii); +22 additional hits |
| 18% | 8/44 | 13 | structural isomerism | 1. same molecular formula but different structural formulae | 9701_w22_qp_22 Q3(a)(ii); 9701_w22_qp_21 Q3(d)(iv); 9701_s21_qp_23 Q5(d); +6 additional hits |
| 18% | 8/44 | 4 | ideal gas equation calculation | 1. use pV = nRT with Pa, m³ and K | 9701_m21_qp_22 Q1(b)(ii); 9701_m21_qp_22 Q1(b)(i); 9701_w20_qp_22 Q3(d)(iii); +3 additional hits |
| 18% | 8/44 | 5 | enthalpy change of formation | 1. enthalpy/energy change when one mole of compound/substance is formed 2. from its elements in their standard states |
9701_w21_qp_22 Q1(b); 9701_s20_qp_21 Q3(a); 9701_m20_qp_22 Q1(g)(ii); +6 additional hits |
| 18% | 8/44 | 8 | temperature effect | 1. higher temperature means particles have greater average kinetic energy 2. the Boltzmann distribution becomes broader with a lower peak and shifts to higher energy 3. a greater proportion of particles have E ≥ Ea 4. frequency of successful/effective collisions increases |
9701_s25_qp_21 Q3(a)(ii); 9701_s24_qp_23 Q2(b)(ii) |
| 16% | 7/44 | 22 | mass spectrometry fragmentation | 1. fragment ions are formed when molecular ions break into smaller positive ions/radicals 2. m/z of a fragment helps identify part of the molecule 3. common hydrocarbon fragments include CH₃⁺ at m/z 15, C₂H₅⁺ at m/z 29 and C₃H₇⁺ at m/z 43, if supported by the question data |
9701_w25_qp_23; 9701_w25_qp_21; 9701_s25_qp_24; 9701_s24_qp_22; +3 additional hits |
| 16% | 7/44 | 12 | photochemical smog / PAN | 1. unburned/unburnt hydrocarbons / VOCs react or combine with NO and/or NO₂ 2. PAN / peroxyacetyl nitrate / peroxyacetylnitrate is formed 3. PAN is a component of photochemical smog |
9701_s25_qp_23 Q3(a)(ii); 9701_s24_qp_23 Q2(d)(ii); 9701_s22_qp_21 Q2(b)(i); 9701_m22_qp_22 Q2(d)(ii); +3 additional hits |
| 16% | 7/44 | 13 | stereoisomerism | 1. same structural formula but different spatial arrangement of atoms/groups | 9701_w22_qp_23 Q5(a)(iii); 9701_w22_qp_21 Q5(a)(iii); 9701_w21_qp_22 Q3(e)(iv); +8 additional hits |
| 16% | 7/44 | 3 | coordinate/dative bond definition | 1. a covalent bond/shared pair is formed 2. both electrons in the shared pair are donated by one atom/species |
9701_s21_qp_21 Q2(a)(i); 9701_m21_qp_22 Q2(e)(i); 9701_s20_qp_21 Q1(b)(ii); +4 additional hits |
| 14% | 6/44 | 8 | Boltzmann distribution curve | 1. label activation energy, Ea, on the energy axis 2. higher temperature: peak is lower, curve is broader and shifted to the right; area under curve stays the same 3. catalyst: draw/mark a lower Ea; the distribution curve itself is unchanged 4. area to the right of Ea represents particles with enough energy for successful collisions |
9701_s25_qp_23; 9701_s24_qp_23; 9701_s22_qp_23; 9701_m22_qp_22; +2 additional hits |
| 14% | 6/44 | 11 | AgNO₃ / NH₃ test | 1. acidify with dilute HNO₃, then add aqueous AgNO₃ 2. Cl⁻ gives a white precipitate, soluble in dilute NH₃ 3. Br⁻ gives a cream/off-white precipitate, partly/slightly soluble in concentrated NH₃ 4. I⁻ gives a yellow precipitate, insoluble in concentrated NH₃ |
9701_w22_qp_21 Q2(c); 9701_s22_qp_22 Q2(d); 9701_m20_qp_22 Q2(c)(i); +6 additional hits |
Formula / Expression Table From Recent P2
统计口径:2020-2025 AS Paper 2 QP/MS,N = 44;本表只收录题面或 MS 中明确出现、要求写出或要求代入的公式/表达式。Frequency 按命中卷数统计;具体来源列的是可直接用文件名复制检索的 QP stem。
| Frequency | Area | Formula / expression to write | MS-ready use | Sources |
|---|---|---|---|---|
| 27% 12/44 |
Kp / Kc overall | General equilibrium: aA + bB ⇌ cC + dD Kc = [C]c[D]d / ([A]a[B]b) Gas equilibrium: aA(g) + bB(g) ⇌ cC(g) + dD(g) Kp = p(C)cp(D)d / (p(A)ap(B)b) One example: H₂(g) + I₂(g) ⇌ 2HI(g): Kc = [HI]² / ([H₂][I₂]); Kp = p(HI)² / (p(H₂)p(I₂)) |
1. [X] means equilibrium concentration of X 2. p(X) means equilibrium partial pressure of gas X 3. powers are the balanced-equation coefficients a, b, c, d 4. omit pure solids/liquids; derive units from the final expression |
CIE_化学AS_A2_计算公式_答案版.xlsx AS rows 13-14; 9701_s23_qp_23 Q4(b)(ii); 9701_w21_qp_22 Q1(c)(i-ii); +10 additional hits |
| 18% 8/44 |
ideal gas | pV = nRT; therefore n = pV / RT, p = nRT / V, and Mr = mass / n | 1. use Pa, m³ and K with R = 8.31 2. convert cm³/dm³/kPa before substitution 3. check whether the question asks for pressure, amount, volume or Mr |
9701_w23_qp_23 Q3(d)(i); 9701_w23_qp_21 Q3(d)(i); 9701_w22_qp_22 Q2(c)(ii); 9701_m21_qp_22 Q1(b)(i-ii); 9701_w20_qp_22 Q3(d)(iii); +3 additional hits |
| 11% 5/44 |
partial pressure | mole fraction = amount of gas / total amount of gas partial pressure = mole fraction × total pressure |
1. calculate mole fraction before partial pressure 2. keep pressure units consistent with the Kp units requested |
9701_s24_qp_23 Q2(c)(ii-iii); 9701_w23_qp_22 Q2(c); 9701_w21_qp_22 Q1(c)(ii); 9701_s21_qp_23 Q2(c)(ii) |
| 11% 5/44 |
bond energy | ΔH = Σ(bond energies broken) - Σ(bond energies formed) | 1. bonds broken are positive 2. bonds formed are subtracted 3. multiply each bond energy by the number of that bond in the balanced equation |
9701_s22_qp_23 Q3(a)(ii); 9701_w21_qp_22 Q1(c)(iii); 9701_m21_qp_22 Q2(c)(i); +3 additional hits |
| 11% 5/44 |
M+1 carbon count | number of C atoms = ([M+1]⁺ abundance / M⁺ abundance) × 100 / 1.1 | 1. use the [M+1]⁺ peak caused mainly by ¹³C 2. rearrange if the [M+1]⁺ abundance is the unknown 3. round to a whole number of carbon atoms |
9701_w25_qp_21 Q4(c)(iii); 9701_s23_qp_23 Q5(c); 9701_s23_qp_21 Q4(b)(iii); +1 additional hits |
| 9% 4/44 |
calorimetry | q = mcΔT ΔH = -q / n |
1. q is heat change of the solution 2. use mass of solution, specific heat capacity and temperature change 3. divide by moles of limiting reagent 4. convert J to kJ mol⁻¹ and use the correct sign |
9701_s23_qp_23 Q3(b)(ii); 9701_s23_qp_21 Q2(b)(ii); 9701_s20_qp_22 Q3(d)(ii); +1 additional hits |
| 9% 4/44 |
water of crystallisation | x = n(H₂O) / n(anhydrous salt) | 1. n(H₂O) = mass lost / 18.0 2. n(anhydrous salt) = mass of residue / Mr 3. simplify the ratio to find x |
9701_s23_qp_21 Q2(b)(ii); 9701_s20_qp_23 Q2(d)(ii); 9701_s20_qp_21 Q2(b); +1 additional hits |
| 2% 1/44 |
M+2 halogen pattern | one Cl: M : M+2 ≈ 3 : 1 one Br: M : M+2 ≈ 1 : 1 |
1. M+2 comes from ³⁷Cl instead of ³⁵Cl, or ⁸¹Br instead of ⁷⁹Br 2. use the ratio to identify Cl/Br in the molecule |
9701_s22_qp_23 Q5(b)(ii) |
Calculation Topic Table / 计算专题
计算专题把“需要代入、换算、判断限制试剂、由数据推出式量/分子式”的题型集中在一起。频率仍按 2020-2025 AS Paper 2 QP/MS,N = 44 统计;其中 molecular formula 和 limiting reagent 属于题干/计算步骤的 text-scan 命中,使用时应回到 Sources 核对题目要求。
| Frequency | Papers | Calculation skill | Formula / method | MS-ready checks | Sources |
|---|---|---|---|---|---|
| 70% | 31/44 | molecular formula from empirical formula / Mr | 1. find empirical formula from mole ratio 2. calculate empirical-formula mass 3. n = Mr(molecular formula) / Mr(empirical formula) 4. multiply every empirical-formula subscript by n |
1. n must be a whole number 2. molecular formula gives the actual number of atoms in one molecule 3. check whether the question gives Mr from mass spectrum, ideal gas or other data |
9701_w25_qp_21; 9701_s25_qp_24; 9701_s25_qp_23; 9701_m25_qp_22; +27 additional hits |
| 27% | 12/44 | Kp / Kc expression and units | For aA + bB ⇌ cC + dD, write products over reactants and raise each concentration/partial pressure to its coefficient. | 1. omit pure solids and liquids 2. use equilibrium values 3. derive units from the final expression after simplification |
9701_s23_qp_23 Q4(b)(ii); 9701_w21_qp_22 Q1(c)(i-ii); +10 additional hits |
| 23% | 10/44 | percentage yield | percentage yield = actual yield / theoretical yield × 100% | 1. theoretical yield is usually calculated from the limiting reagent 2. compare masses with masses, or moles with moles, consistently 3. answer cannot normally exceed 100% unless the sample is impure/wet |
9701_s25_qp_21; 9701_s24_qp_23; 9701_s24_qp_21; 9701_s23_qp_22; 9701_m23_qp_22; +5 additional hits |
| 20% | 9/44 | limiting reagent | 1. convert each reactant amount to moles 2. divide by its balanced-equation coefficient, or calculate the amount of product each reactant can form 3. the reactant producing the smaller product amount is limiting |
1. use the balanced equation 2. excess reagent remains unreacted 3. final yield must be based on the limiting reagent, not the reagent with the larger mass |
9701_s25_qp_23; 9701_s25_qp_22; 9701_s22_qp_22; 9701_w21_qp_23; 9701_w21_qp_21; +4 additional hits |
| 18% | 8/44 | ideal gas equation | pV = nRT; rearrange to find n, p, V or Mr = mass / n. | 1. use Pa, m³ and K with R = 8.31 2. convert cm³/dm³/kPa before substitution 3. check whether gas amount is linked to a balanced equation |
9701_w23_qp_23 Q3(d)(i); 9701_w23_qp_21 Q3(d)(i); 9701_w22_qp_22 Q2(c)(ii); +5 additional hits |
| 11% | 5/44 | partial pressure / mole fraction | mole fraction = amount of gas / total amount of gas partial pressure = mole fraction × total pressure |
1. calculate all gaseous amounts at equilibrium if the reaction changes mole numbers 2. keep pressure units consistent with Kp units |
9701_s24_qp_23 Q2(c)(ii-iii); 9701_w23_qp_22 Q2(c); 9701_w21_qp_22 Q1(c)(ii); 9701_s21_qp_23 Q2(c)(ii) |
| 11% | 5/44 | bond energy enthalpy | ΔH = Σ(bond energies broken) - Σ(bond energies formed) | 1. multiply by the number of each bond in the balanced equation 2. bonds broken are positive; bonds formed are subtracted 3. final unit is usually kJ mol⁻¹ |
9701_s22_qp_23 Q3(a)(ii); 9701_w21_qp_22 Q1(c)(iii); 9701_m21_qp_22 Q2(c)(i); +3 additional hits |
| 11% | 5/44 | M+1 carbon count | number of C atoms = ([M+1]⁺ abundance / M⁺ abundance) × 100 / 1.1 | 1. [M+1]⁺ is mainly from ¹³C 2. round to a whole number of carbon atoms 3. combine with Mr/IR/NMR information if supplied |
9701_w25_qp_21 Q4(c)(iii); 9701_s23_qp_23 Q5(c); 9701_s23_qp_21 Q4(b)(iii); +1 additional hits |
| 9% | 4/44 | calorimetry | q = mcΔT ΔH = -q / n |
1. q is heat change of the solution 2. divide by moles of limiting reagent 3. convert J to kJ mol⁻¹ and choose sign from exothermic/endothermic context |
9701_s23_qp_23 Q3(b)(ii); 9701_s23_qp_21 Q2(b)(ii); 9701_s20_qp_22 Q3(d)(ii); +1 additional hits |
| 9% | 4/44 | hydrated salt / water of crystallisation | 1. n(H₂O) = mass lost / 18.0 2. n(anhydrous salt) = mass of residue / Mr 3. x = n(H₂O) / n(anhydrous salt) |
1. use mass lost for water 2. simplify the H₂O : salt ratio 3. check whether the salt formula already contains more than one formula unit |
9701_s23_qp_21 Q2(b)(ii); 9701_s20_qp_23 Q2(d)(ii); 9701_s20_qp_21 Q2(b); +1 additional hits |
High-Frequency Colour / State / Precipitate Observations
频率口径:只统计 2020-2025 AS Paper 2 MS 中明确作为 observation / appearance / state / precipitate 给分的答案片段;同一份卷子同一现象只计 1 次。
| Frequency | Papers | Chapter | Observation / substance | MS-ready wording | Sources |
|---|---|---|---|---|---|
| 30% | 13/44 | 14 / 17 | Br₂(aq) / bromine water | 1. alkene / C=C: orange/brown/red bromine water turns colourless / decolourises 2. no C=C: no visible change / solution stays orange-brown |
9701_s21_qp_23 Q1(b); 9701_s21_qp_21 Q5(c)(ii); 9701_w20_qp_22 Q4(c); +11 additional hits |
| 30% | 13/44 | 17 | 2,4-DNPH | 1. aldehydes and ketones give a red/orange/yellow precipitate / solid / crystals 2. no carbonyl compound: no precipitate / no visible change |
9701_s21_qp_23 Q4(b); 9701_w20_qp_22 Q4(c); 9701_s20_qp_21 Q6(d); +10 additional hits |
| 20% | 9/44 | 11 | halogen colours and states | 1. chlorine: green / yellow-green gas 2. bromine: orange/brown/red-brown liquid 3. iodine: dark grey/black solid; iodine vapour is purple |
9701_w24_qp_23 Q2(a)(i); 9701_w24_qp_21 Q2(a)(i); 9701_s21_qp_23 Q1(a)(ii); +8 additional hits |
| 18% | 8/44 | 14 / 18 | carbonate / hydrogencarbonate with acid | 1. effervescence / fizzing / bubbling is seen 2. gas is CO₂; if tested, CO₂ turns limewater milky 3. no carboxylic acid/acidic group: no visible change |
9701_s22_qp_21 Q4(b)(i); 9701_s21_qp_23 Q4(b); 9701_w20_qp_22 Q4(c); +5 additional hits |
| 16% | 7/44 | 16 / 17 | alkaline I₂ / iodoform test | 1. yellow precipitate forms 2. precipitate is CHI₃ / iodoform / triiodomethane 3. positive for CH₃CO- or CH₃CH(OH)- groups |
9701_w22_qp_23 Q4(a); 9701_w22_qp_22 Q3(a)(i); 9701_w22_qp_21 Q4(a); +4 additional hits |
| 16% | 7/44 | 17 | Tollens’ / Fehling’s observations | 1. Tollens’ reagent: aldehyde gives silver mirror or silver/grey/black precipitate 2. Fehling’s solution: aldehyde gives brick-red precipitate 3. ketone: no reaction / no visible change |
9701_m22_qp_22 Q4(a)(i); 9701_m21_qp_22 Q4(b); 9701_w20_qp_22 Q4(a)(iv); +4 additional hits |
| 16% | 7/44 | 11 / 15 | AgNO₃ halide precipitates | 1. Cl⁻ gives white AgCl precipitate 2. Br⁻ gives cream/off-white AgBr precipitate 3. I⁻ gives yellow AgI precipitate |
9701_w23_qp_23 Q1(c); 9701_w23_qp_21 Q1(c); 9701_s22_qp_22 Q2(d); +4 additional hits |
| 14% | 6/44 | 11 / 15 | AgX precipitate solubility in NH₃ | 1. AgCl dissolves in dilute/excess NH₃ 2. AgBr is partly/slightly soluble in concentrated NH₃ 3. AgI is insoluble; solid remains |
9701_s22_qp_22 Q2(d); 9701_s20_qp_22 Q4(a)(iii); 9701_m20_qp_22 Q2(c)(i); +3 additional hits |
| 14% | 6/44 | 16 / 18 | sodium metal test | 1. Na(s) disappears/dissolves and effervescence / fizzing / bubbles are seen 2. gas is H₂ 3. positive with alcohols or carboxylic acids; sodium oxide gives no effervescence |
9701_s23_qp_23 Q5(d); 9701_w22_qp_23 Q4(a); 9701_w22_qp_21 Q4(a); +3 additional hits |
| 9% | 4/44 | 16 / 17 | acidified K₂Cr₂O₇ / KMnO₄ colour change | 1. acidified K₂Cr₂O₇ changes from orange to green; Cr³⁺ is green 2. acidified KMnO₄ / MnO₄⁻ changes from purple to colourless 3. no oxidation: no visible change |
9701_w22_qp_23 Q4(a); 9701_w22_qp_22 Q3(a)(i); 9701_w22_qp_21 Q4(a); +1 additional hits |
| 7% | 3/44 | 11 | concentrated H₂SO₄ with halides | 1. Cl⁻: steamy/misty fumes of HCl; acid-base only 2. Br⁻: brown/red-brown Br₂ forms; H₂SO₄ is reduced 3. I⁻: black solid / purple vapour I₂, yellow sulfur, rotten-egg H₂S and/or effervescence may be seen |
9701_w25_qp_24 Q1(c)(i); 9701_w23_qp_23 Q1(c); 9701_w23_qp_21 Q1(c) |
| 7% | 3/44 | 9 / 10 | sulfate / hydroxide precipitates | 1. BaSO₄ is an insoluble white precipitate/solid 2. Mg(OH)₂ is insoluble / forms a white precipitate; Ba(OH)₂ is soluble enough to distinguish 3. use a soluble sulfate or hydroxide reagent when asked |
9701_s23_qp_21 Q2(c)(ii); 9701_m23_qp_22 Q2(c); 9701_s22_qp_21 Q1(c) |
| 7% | 3/44 | 9 | Period 3 chloride states / water observations | 1. SiCl₄ is a colourless liquid; with water it gives misty fumes and white solid/suspension/precipitate of SiO₂ 2. PCl₅ is a white/pale yellow solid; with water it gives misty/steamy fumes and an acidic colourless solution 3. MgCl₂/PCl₅ may be white crystals/solid; SiCl₄ is the key liquid exception |
9701_s25_qp_22 Q1(b)(ii); 9701_s23_qp_21 Q3(b); 9701_s21_qp_23 Q3(a)(i); +1 additional hits |
| 0% | 0/44 | 12 | nitrogen oxides appearance | 1. NO is a colourless gas 2. NO₂ is a brown gas 3. this is syllabus/textbook background; no recent P2 MS observation hit in the current 44-paper set |
no 2020-2025 P2 MS hit |
Organic Mechanism MS Screenshots
机理题按文字采分点背;截图只用来核对 curly arrow 起点、终点、中间体和电荷位置。
| Frequency | Papers | Chapter | Mechanism | MS screenshot | Sources |
|---|---|---|---|---|---|
| 64% | 28/44 | 14-17 | electrophilic addition to C=C / curly-arrow rule 1. curly arrow starts at the C=C π bond 2. show induced dipole in Br₂ / H-X and bond-breaking arrow to the leaving atom 3. draw the carbocation / cyclic intermediate as required 4. nucleophile arrow starts from lone pair/negative charge |
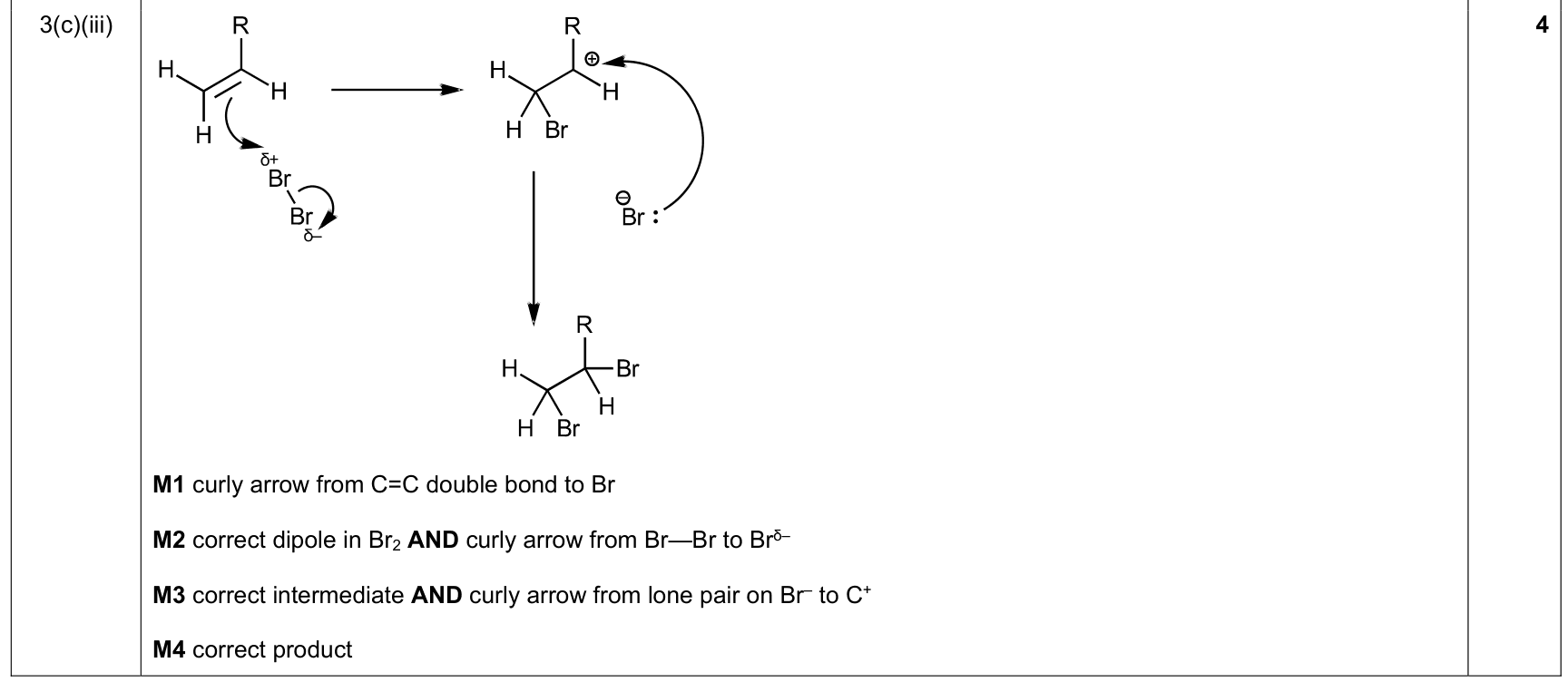 |
9701_s25_qp_23 Q6(b); 9701_s25_qp_22 Q3(a)(ii); 9701_w24_qp_22 Q4(a); +25 additional hits |
| 18% | 8/44 | 14 | free-radical substitution 1. initiation: homolytic fission in UV light forms halogen radicals 2. propagation 1: a halogen radical removes H from an alkane to form an alkyl radical and HCl/HBr 3. propagation 2: the alkyl radical reacts with Cl2/Br2 to form a halogenoalkane and regenerate a halogen radical 4. termination: two radicals combine to form a stable molecule |
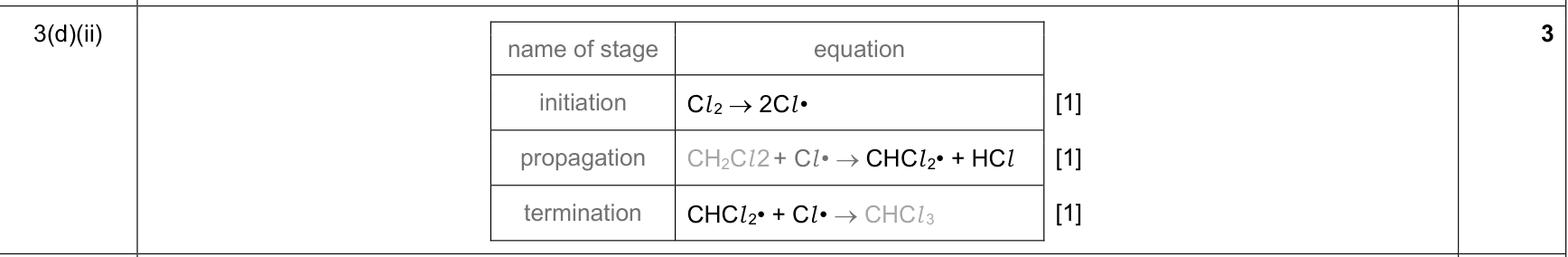 |
9701_w24_qp_21 Q2(b)(iii); 9701_m21_qp_22 Q2(g)(i); 9701_s20_qp_23 Q4(b)(ii); +22 additional hits |
| 9% | 4/44 | 15 | nucleophilic substitution of tertiary/secondary halogenoalkane (SN1-style shown) 1. show Cδ+–Xδ− dipole 2. curly arrow from C-X bond to X when bond breaks 3. draw the carbocation / intermediate where required 4. curly arrow from nucleophile lone pair to C+ / electron-deficient carbon |
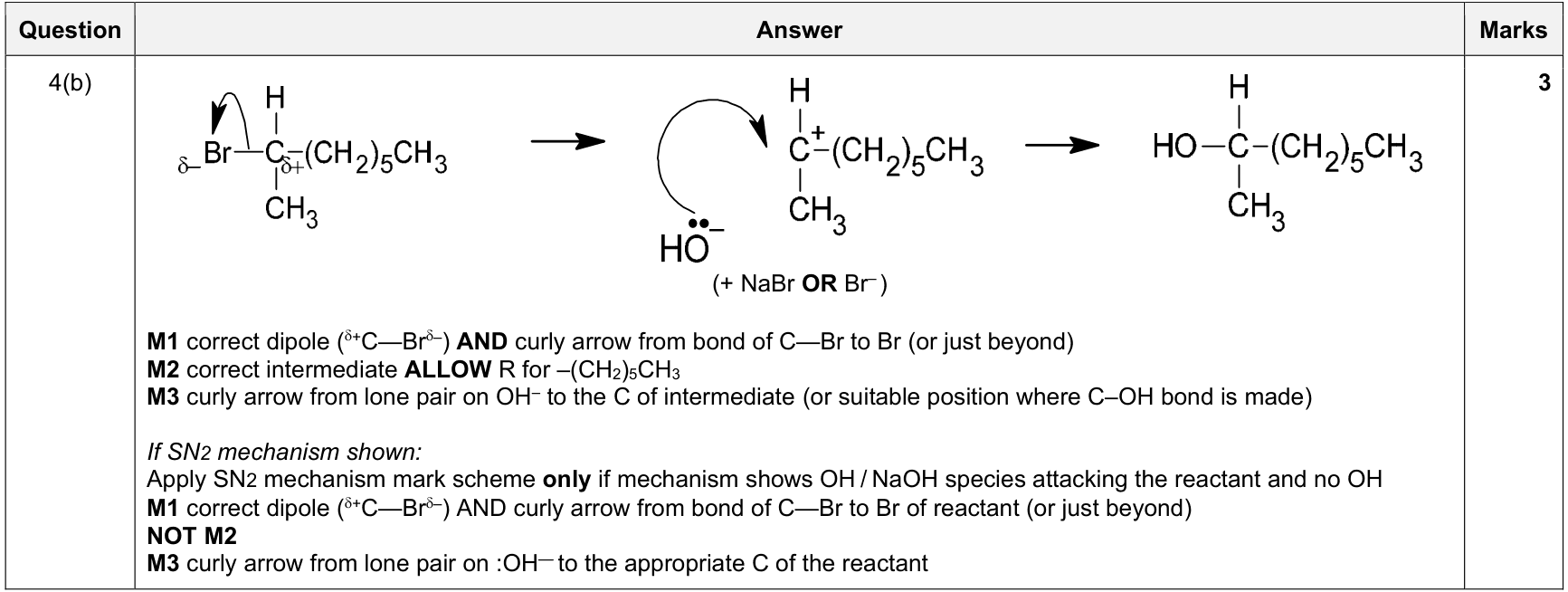 |
9701_s24_qp_22 Q4(b); 9701_s21_qp_22 Q3(c)(i); 9701_w20_qp_21 Q4(c)(ii); +2 additional hits |
| 2% | 1/44 | 15 / 19 | SN2 nucleophilic substitution / CN⁻ chain extension 1. show Cδ+–Brδ− dipole 2. curly arrow from C–Br bond to Br / Brδ− as bond breaks 3. curly arrow starts from lone pair on C of CN⁻ 4. arrow goes to C-1 / δ⁺ carbon of the bromoalkane; no carbocation intermediate is drawn |
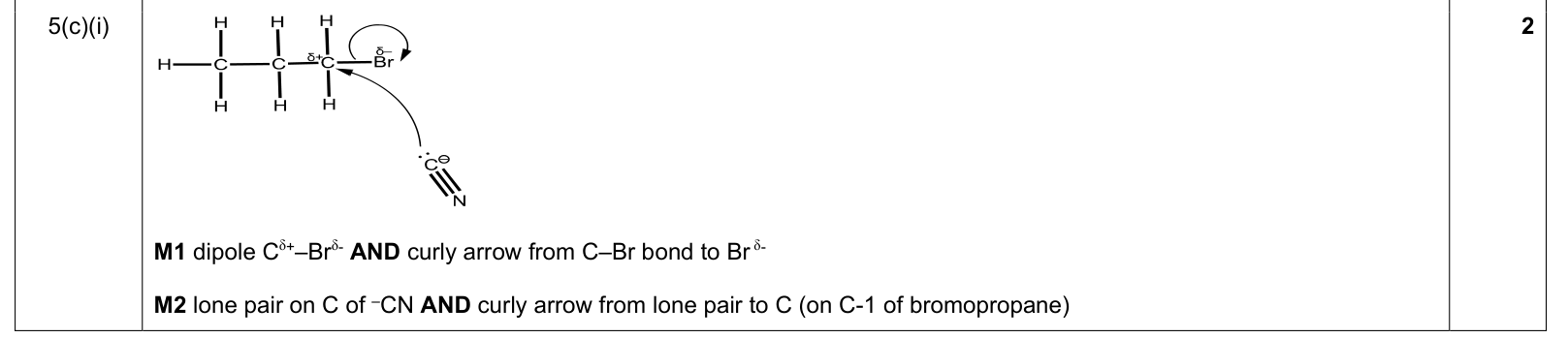 |
9701_s22_qp_22 Q5(c)(i) |
| 9% | 4/44 | 17 / 19 | HCN nucleophilic addition to carbonyl 1. curly arrow from lone pair on CN⁻ to δ⁺ carbonyl carbon 2. show C=O dipole and arrow from C=O π bond to O 3. protonation arrow from O⁻/alkoxide to H of HCN/H⁺; regenerate CN⁻ if shown |
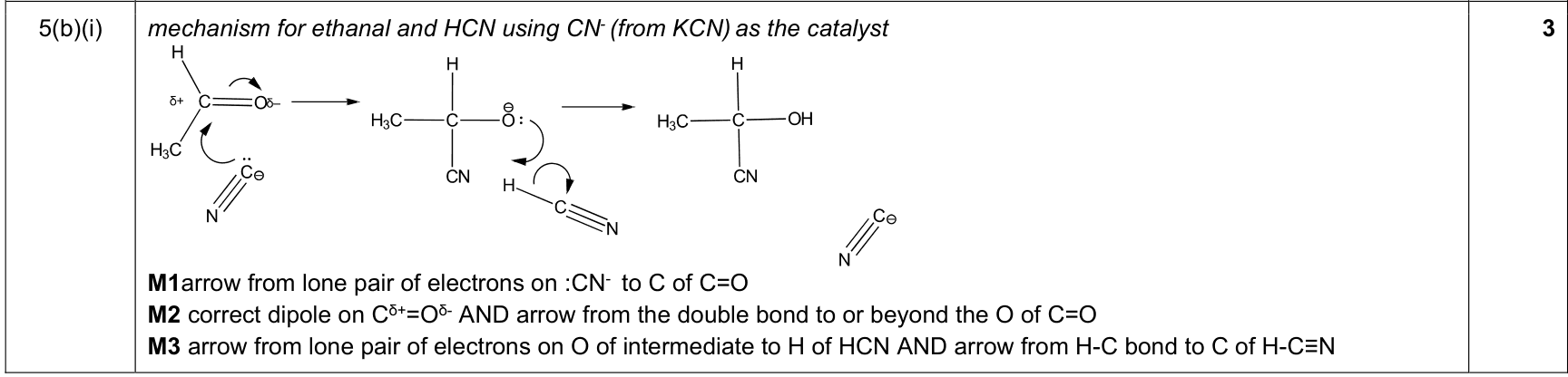 |
9701_s23_qp_23 Q6(b)(ii); 9701_w20_qp_21 Q3(c)(iii); 9701_s20_qp_22 Q5(b)(i); +8 additional hits |
Cross-Chapter MS Answering Rules
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 64% | 28/44 | 28 | mechanism curly-arrow rule | 1. curly arrows start at a lone pair/electron pair/bond/negative charge 2. arrow points to the atom or bond receiving the electron pair 3. for bond breaking, arrow goes from the bond to the atom receiving the electrons |
9701_w25_qp_24 Q3(b)(ii); 9701_s25_qp_22 Q3(a)(ii); 9701_m25_qp_22 Q4(b)(i); +25 additional hits |
| 36% | 16/44 | 19 | equations / state symbols | 1. use correct formulae 2. balance the equation 3. include charges where needed 4. include state symbols when the question asks for them |
9701_s20_qp_22 Q1(e)(ii); 9701_s20_qp_21 Q2(a)(i); 9701_s20_qp_21 Q1(c)(i); +20 additional hits |
| 34% | 15/44 | 15 | organic conditions | 1. acidified K₂Cr₂O₇/KMnO₄ plus distil/reflux 2. KCN/NaOH in ethanol plus heat/reflux 3. NH₃ in ethanol heated under pressure 4. conditions are often separate marks |
9701_s20_qp_22 Q4(a)(iii); 9701_m20_qp_22 Q3(a)(i); 9701_m20_qp_22 Q2(c)(i); +38 additional hits |
1 Atomic Structure / Electrons In Atoms
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 36% | 16/44 | 18 | IE trend explanation | 1. compare nuclear charge/proton number 2. compare distance/atomic radius and shielding by inner shells or sub-shells 3. stronger attraction for the outer electron gives a higher ionisation energy 4. exceptions: Al is lower than Mg because its electron is removed from 3p rather than 3s; S is lower than P because paired 3p electrons repel each other (spin-pair repulsion) |
9701_w20_qp_22 Q1(b)(iv); 9701_w20_qp_21 Q1(c); 9701_w20_qp_21 Q1(b); +24 additional hits |
| 20% | 9/44 | 9 | ionic radius / isoelectronic comparison | 1. compare the number of occupied electron shells first 2. in an isoelectronic series, ions have the same number of electrons / same electron arrangement 3. the ion with the greater nuclear charge / more protons has a smaller ionic radius because the same electron cloud is attracted more strongly 4. down a group, ionic radius increases because there are more occupied shells and more shielding |
9701_w25_qp_23; 9701_w25_qp_21; 9701_s25_qp_24; 9701_s25_qp_23; 9701_m25_qp_22; +4 additional hits |
| 9% | 4/44 | 4 | same chemical properties of isotopes | 1. same electronic configuration 2. same number and arrangement of outer-shell electrons |
9701_s25_qp_21 Q2(e); 9701_s22_qp_23 Q1(d) |
| 7% | 3/44 | 3 | free radical definition | 1. species with one or more unpaired electrons | 9701_w24_qp_23 Q2(b)(i); 9701_w24_qp_21 Q2(b)(i); 9701_w23_qp_22 Q2(e)(i) |
| 7% | 3/44 | 3 | isotopes definition | 1. same number of protons 2. different number of neutrons |
9701_s22_qp_21 Q1(e)(iii); 9701_s22_qp_21 Q1(e)(ii); 9701_s20_qp_21 Q1(a)(ii); +7 additional hits |
| 2% | 1/44 | 1 | successive ionisation equation | 1. write Xⁿ⁺(g) → X⁽ⁿ⁺¹⁾⁺(g) + e⁻ 2. one electron is removed from a gaseous ion 3. state symbols are needed 常写方程 / Fixed equation知识点归属X(g) → X⁺(g) + e⁻
Xⁿ⁺(g) → X⁽ⁿ⁺¹⁾⁺(g) + e⁻ 使用场景:1. include gaseous state symbols for atom/ion
2. show one electron removed 3. use the correct charge for the numbered IE |
9701_s25_qp_24 Q1(c)(i); 9701_m23_qp_22 Q2(a)(i) |
2 Atoms, Molecules And Stoichiometry
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 20% | 9/44 | 9 | relative atomic/isotopic/molecular mass | 1. relative isotopic mass = mass of one atom of an isotope compared with 1/12 of the mass of a carbon-12 atom 2. relative atomic mass = weighted mean mass of atoms of an element compared with 1/12 of carbon-12 3. relative molecular/formula mass = sum of the relative atomic masses in the molecule/formula unit |
9701_w22_qp_21 Q1(a)(ii); 9701_s20_qp_23 Q2(a); 9701_s20_qp_22 Q1(a); +6 additional hits |
| 9% | 4/44 | 4 | empirical formula definition | 1. empirical formula = simplest/lowest whole-number ratio of atoms of each element in a compound 2. molecular formula = actual number of atoms of each element in one molecule 3. molecular formula is obtained from empirical formula using n = Mr(molecular formula) / Mr(empirical formula) |
9701_s22_qp_21 Q4(a); 9701_s21_qp_23 Q3(c); 9701_s20_qp_23 Q3(b)(iii); +1 additional hits |
| 9% | 4/44 | 4 | hydrated salt / water of crystallisation calculation | 1. calculate mol anhydrous salt 2. calculate mol H₂O separately 3. find the simplest H₂O : salt ratio |
9701_s23_qp_21 Q2(b)(ii); 9701_s20_qp_23 Q2(d)(ii); 9701_s20_qp_21 Q2(b); +1 additional hits |
| 7% | 3/44 | 3 | mole / Avogadro constant | 1. one mole contains Avogadro constant / 6.02 × 10²³ specified particles 2. number of particles = amount in mol × Avogadro constant 3. state the particles being counted: atoms, molecules, ions or electrons |
9701_s25_qp_23 Q4(a)(iv); 9701_s22_qp_23 Q1(f)(i); 9701_s21_qp_21 Q1(a)(iii) |
3 Chemical Bonding
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 59% | 26/44 | 23 | bonding diagrams: metallic / dot-and-cross / coordinate | 1. for dot-and-cross diagrams, show outer-shell electrons only and use different symbols for electrons from different atoms 2. covalent bonds are shared pairs; coordinate/dative bonds have both electrons in the shared pair donated by one atom/species 3. metallic bonding diagrams need a lattice/regular arrangement of positive ions/cations and delocalised electrons 4. for NH₄Cl, show ionic attraction between NH₄⁺ and Cl⁻, four N-H covalent bonds in NH₄⁺, and one N→H coordinate bond / one N-H bonding pair from N |
9701_s24_qp_23 Q4(a); 9701_w20_qp_21 Q3(a)(i); 9701_s20_qp_22 Q1(b); +23 additional hits |
| 50% | 22/44 | 27 | simple molecular boiling/melting | 1. greater number of electrons in the molecules 2. stronger instantaneous dipole-induced dipole forces / London dispersion forces / van der Waals’ forces between molecules 3. instantaneous dipoles induce dipoles in neighbouring molecules, producing attraction between δ⁺ and δ⁻ ends 4. more energy is needed to overcome the stronger intermolecular forces |
9701_w20_qp_23 Q4(a)(iii); 9701_w20_qp_21 Q4(a)(iii); 9701_s20_qp_21 Q1(b)(i); +21 additional hits |
| 43% | 19/44 | 14 | σ and π bonds / orbital overlap / hybridisation | 1. a σ bond is formed by direct/head-on/end-on overlap of orbitals between the bonding atoms 2. a π bond is formed by sideways/lateral overlap of adjacent p orbitals 3. single bond = 1σ; double bond = 1σ + 1π; triple bond = 1σ + 2π 4. common hybridisation: sp gives linear/triple-bond centres; sp² gives trigonal planar/C=C centres; sp³ gives tetrahedral/saturated carbon or ammonium centres |
9701_s24_qp_23 Q1(a)(ii); 9701_s24_qp_21 Q4(b)(i-ii); 9701_s22_qp_21 Q2(a); +16 additional hits |
| 32% | 14/44 | 14 | metallic bonding / conductivity | 1. giant metallic lattice / regular arrangement of positive ions/cations surrounded by delocalised electrons 2. many strong electrostatic attractions between cations and delocalised electrons / strong metallic bonds need more energy to overcome, so melting point is high 3. delocalised electrons are free to move through the structure and carry charge |
9701_w24_qp_21 Q3(a)(iii); 9701_s22_qp_23 Q1(f)(ii); 9701_s22_qp_22 Q1(a); +5 additional hits |
| 16% | 7/44 | 7 | coordinate/dative bond definition | 1. a covalent bond/shared pair is formed 2. both electrons in the shared pair are donated by one atom/species |
9701_s21_qp_21 Q2(a)(i); 9701_m21_qp_22 Q2(e)(i); 9701_s20_qp_21 Q1(b)(ii); +4 additional hits |
| 14% | 6/44 | 6 | hydrogen bonding | 1. H atom is bonded to highly electronegative N/O/F in one molecule 2. H atom is attracted to a lone pair on N/O/F in another molecule 3. hydrogen bonds are stronger than other intermolecular forces, so more energy is needed to overcome them |
9701_w21_qp_22 Q1(a)(ii); 9701_w21_qp_21 Q4(d); 9701_s21_qp_23 Q2(b)(iii); +3 additional hits |
| 9% | 4/44 | 4 | covalent bond definition | 1. electrostatic attraction 2. between the nuclei of two atoms and a shared pair of electrons |
9701_w21_qp_22 Q2(d)(iii); 9701_s21_qp_22 Q2(b)(i); 9701_s21_qp_21 Q2(a)(i); +13 additional hits |
| 5% | 2/44 | 2 | electronegativity definition | 1. power of an atom to attract a bonding pair / shared pair of electrons to itself | 9701_w24_qp_22 Q2(c)(ii); 9701_s24_qp_23 Q4(c)(ii); 9701_m22_qp_22 Q3(a); +1 additional hits |
| 27% | 12/44 | 12 | bond polarity / permanent dipole forces | 1. a polar bond forms when bonded atoms have different electronegativities 2. the more electronegative atom attracts the bonding pair more strongly and becomes δ⁻; the other atom becomes δ⁺ 3. permanent dipole-permanent dipole forces act between polar molecules when δ⁺ and δ⁻ ends attract |
9701_w25_qp_23; 9701_w25_qp_21; 9701_s25_qp_22; 9701_m25_qp_22; 9701_w24_qp_22; +7 additional hits |
| 5% | 2/44 | 2 | ionic lattice melting point | 1. many strong electrostatic attractions between oppositely charged ions 2. more energy is needed to overcome them |
9701_s22_qp_23 Q2(b)(ii); 9701_w20_qp_23 Q3(a)(iv); 9701_w20_qp_21 Q3(a)(iv); +2 additional hits |
| 9% | 4/44 | 4 | VSEPR shape / bond angle | 1. electron pairs repel 2. lone pairs repel more than bond pairs 3. give the correct shape and bond angle |
9701_w20_qp_22 Q2(f); 9701_s20_qp_22 Q1(d); 9701_s20_qp_21 Q4(c); +12 additional hits |
4 States Of Matter
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 18% | 8/44 | 8 | ideal gas equation calculation | 1. use pV = nRT with Pa, m³ and K | 9701_m21_qp_22 Q1(b)(ii); 9701_m21_qp_22 Q1(b)(i); 9701_w20_qp_22 Q3(d)(iii); +3 additional hits |
| 5% | 2/44 | 2 | temperature and kinetic energy | 1. higher temperature means greater average kinetic energy | 9701_m21_qp_22 Q1(b)(iii) |
| 2% | 1/44 | 1 | gas pressure explanation | 1. pressure is force per unit area from collisions of gas molecules with the container walls | 9701_s21_qp_23 Q2(b)(i); 9701_m21_qp_22 Q3(d)(iv); 9701_m21_qp_22 Q1(b)(iii) |
5 Chemical Energetics
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 18% | 8/44 | 8 | enthalpy change of formation | 1. enthalpy/energy change when one mole of compound/substance is formed 2. from its elements in their standard states 形成焓方程模板 / Formation equation知识点归属elements in standard states → 1 mol compound
Example: H₂(g) + ½O₂(g) → H₂O(l) 使用场景:1. product coefficient must be 1
2. reactants are elements in their standard states 3. include state symbols when the question/MS asks |
9701_w21_qp_22 Q1(b); 9701_s20_qp_21 Q3(a); 9701_m20_qp_22 Q1(g)(ii); +6 additional hits |
| 11% | 5/44 | 5 | bond energy calculation | 1. ΔH = sum of bond energies broken - sum of bond energies formed | 9701_s22_qp_23 Q3(a)(ii); 9701_w21_qp_22 Q1(c)(iii); 9701_m21_qp_22 Q2(c)(i); +3 additional hits |
| 9% | 4/44 | 4 | enthalpy change of combustion | 1. enthalpy/energy change when one mole of substance/compound burns completely in excess O₂有机物燃烧通式 / Combustion equation知识点归属CₓHᵧOᶻ + O₂ → xCO₂ + y/2H₂O
使用场景:1. complete combustion gives CO₂ and H₂O
2. fractional O₂ coefficients are acceptable unless whole numbers are required |
9701_w21_qp_21 Q1(b)(i); 9701_s20_qp_23 Q3(c)(i); 9701_s20_qp_22 Q3(d)(i); +1 additional hits |
| 9% | 4/44 | 4 | q = mcΔT / calorimetry | 1. q = mcΔT for the solution 2. ΔH = -q / moles of limiting reagent 3. convert J to kJ mol⁻¹ 4. include the correct sign |
9701_s23_qp_23 Q3(b)(ii); 9701_s23_qp_21 Q2(b)(ii); 9701_s20_qp_22 Q3(d)(ii); +1 additional hits |
6 Electrochemistry: Redox
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 32% | 14/44 | 15 | reducing agent | 1. donates electrons and is oxidised | 9701_w20_qp_22 Q4(b)(iii); 9701_w20_qp_21 Q3(c)(ii); 9701_m20_qp_22 Q2(c)(ii); +14 additional hits |
| 25% | 11/44 | 13 | oxidising agent | 1. accepts electrons and is reduced | 9701_m21_qp_22 Q4(c)(i); 9701_m20_qp_22 Q2(c)(ii); 9701_m20_qp_22 Q2(b)(i); +11 additional hits |
| 14% | 6/44 | 6 | disproportionation definition | 1. same species is both oxidised and reduced | 9701_s21_qp_21 Q1(b)(ii); 9701_m21_qp_22 Q2(d)(i); 9701_s20_qp_22 Q2(c)(ii); +5 additional hits |
| 11% | 5/44 | 5 | redox by oxidation numbers | 1. one species increases oxidation number while another decreases | 9701_w21_qp_22 Q1(d)(ii); 9701_w21_qp_21 Q1(c)(iii); 9701_s21_qp_21 Q4(a); +2 additional hits |
| 2% | 1/44 | 1 | electron transfer explanation | 1. oxidation is electron loss 2. reduction is electron gain |
9701_m24_qp_22 Q1(d)(i) |
7 Equilibria
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 25% | 11/44 | 11 | Bronsted acid / strong vs weak acid | 1. acid = proton/H⁺ donor 2. strong acid fully dissociates/fully ionises in aqueous solution/water 3. weak acid partially dissociates/partially ionises in aqueous solution/water |
9701_w20_qp_23 Q2(b)(i); 9701_w20_qp_22 Q3(d)(ii); 9701_w20_qp_21 Q2(b)(i); +11 additional hits |
| 14% | 6/44 | 6 | Kp expression / units | 1. Kp = product partial pressures over reactant partial pressures 2. each partial pressure is raised to its stoichiometric coefficient 3. omit solids from the expression 4. derive units from the expression |
9701_w21_qp_22 Q1(c)(i-ii); 9701_s21_qp_23 Q2(c)(iii); 9701_m20_qp_22 Q1(f)(ii); +3 additional hits |
| 14% | 6/44 | 6 | Kc expression / units | 1. Kc = product concentrations over reactant concentrations in square brackets 2. each concentration is raised to its stoichiometric coefficient 3. derive units from the expression 4. pressure/catalyst changes do not change Kc; temperature changes can change Kc |
9701_s24_qp_21 Q3(c)(ii); 9701_s23_qp_23 Q4(b)(ii); 9701_s22_qp_23 Q3(a)(iii); +3 additional hits |
| 11% | 5/44 | 5 | dynamic equilibrium definition | 1. system is closed 2. rate of forward reaction equals rate of reverse/backward reaction 3. concentrations or macroscopic properties remain constant |
9701_w25_qp_21 Q1(d)(i); 9701_s25_qp_21 Q3(b); 9701_s22_qp_22 Q3(a)(i); +2 additional hits |
| 9% | 4/44 | 4 | closed system condition | 1. closed/sealed system or container 2. no substances enter or leave |
9701_w25_qp_22 Q2(e)(ii); 9701_w25_qp_21 Q1(d)(ii); 9701_m20_qp_22 Q1(f)(i); +1 additional hits |
| 5% | 2/44 | 2 | Le Chatelier principle | 1. if a condition changes, the equilibrium shifts/moves to minimise the change | 9701_w25_qp_21 Q1(d)(iii); 9701_s25_qp_21 Q3(b); 9701_s24_qp_21 Q3(a); +1 additional hits |
| 2% | 1/44 | 1 | catalyst and equilibrium | 1. catalyst gives no change in equilibrium position 2. it reduces the time taken to reach the same equilibrium |
9701_s22_qp_23 Q3(a)(iv) |
8 Reaction Kinetics
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 20% | 9/44 | 10 | frequency of effective collisions | 1. rate increases/decreases with the frequency of successful/effective collisions per unit time | 9701_s22_qp_21 Q1(g); 9701_m22_qp_22 Q2(b)(iii); 9701_m21_qp_22 Q1(c)(ii); +7 additional hits |
| 18% | 8/44 | 8 | temperature effect | 1. higher temperature means particles have greater average kinetic energy 2. the Boltzmann distribution becomes broader with a lower peak and shifts to higher energy 3. a greater proportion of particles have E ≥ Ea 4. frequency of successful/effective collisions increases |
9701_s25_qp_21 Q3(a)(ii); 9701_s24_qp_23 Q2(b)(ii) |
| 11% | 5/44 | 5 | catalyst lowers Ea | 1. catalyst provides an alternative route with lower activation energy 2. the Maxwell-Boltzmann curve is not shifted by the catalyst 3. a greater proportion of particles have E ≥ Ea because the activation energy line is lower 4. successful collisions are more frequent |
9701_m21_qp_22 Q1(a)(ii); 9701_s20_qp_21 Q3(e)(i); 9701_m20_qp_22 Q1(b); +7 additional hits |
| 14% | 6/44 | 6 | Boltzmann distribution curve | 1. label activation energy, Ea, on the energy axis 2. higher temperature: peak is lower, curve is broader and shifted to the right; area under curve stays the same 3. catalyst: draw/mark a lower Ea; the distribution curve itself is unchanged 4. area to the right of Ea represents particles with enough energy for successful collisions |
9701_s25_qp_23; 9701_s24_qp_23; 9701_s22_qp_23; 9701_m22_qp_22; 9701_m21_qp_22; 9701_s20_qp_23 |
| 2% | 1/44 | 1 | activation energy definition | 1. minimum energy particles must have for a successful/effective collision | 9701_s20_qp_21 Q3(e)(i); 9701_s20_qp_21 Q3(c); 9701_m20_qp_22 Q1(c); +14 additional hits |
| 2% | 1/44 | 1 | concentration/pressure effect | 1. more particles per unit volume 2. higher frequency of successful/effective collisions |
9701_s25_qp_23 Q4(a)(iii) |
9 Periodicity: Period 3
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 32% | 14/44 | 18 | Period 3 melting point structure | 1. identify the structure: simple molecular / giant metallic / giant covalent as appropriate 2. simple molecular substances: only weak intermolecular / van der Waals’ forces are overcome, so less energy is needed 3. giant metallic/covalent substances: many strong metallic/covalent bonds or attractions must be overcome/broken, so more energy is needed |
9701_w21_qp_23 Q3(a)(i); 9701_w21_qp_21 Q3(a)(i); 9701_w20_qp_22 Q2(f); +12 additional hits |
| 20% | 9/44 | 10 | Period 3 chloride hydrolysis | 1. SiCl₄ + 2H₂O → SiO₂ + 4HCl 2. PCl₅ + 4H₂O → H₃PO₄ + 5HCl 3. include state symbols / acidic products if the question asks Period 3 水解/氧化物方程知识点归属P₄O₁₀ + 6H₂O → 4H₃PO₄
P₄O₁₀ + 12NaOH → 4Na₃PO₄ + 6H₂O PCl₅ + 4H₂O → H₃PO₄ + 5HCl POCl₃ + 3H₂O → H₃PO₄ + 3HCl SiCl₄ + 2H₂O → SiO₂ + 4HCl 使用场景:1. chloride hydrolysis answers need acidic products
2. SiCl₄ gives SiO₂ + HCl in recent MS wording 3. P₄O₁₀ with excess NaOH gives phosphate salt |
9701_w21_qp_23 Q3(b)(ii); 9701_w21_qp_21 Q3(b)(ii); 9701_w20_qp_22 Q3(b)(ii); +12 additional hits |
| 14% | 6/44 | 6 | Period 3 atomic radius | 1. nuclear charge/proton number increases across the period 2. shielding is similar 3. attraction for outer electrons increases, so atomic radius decreases |
9701_w20_qp_22 Q1(b)(iv) |
| 14% | 6/44 | 6 | Period 3 conductivity | 1. conductivity requires mobile charged particles 2. Na, Mg and Al conduct because delocalised electrons are free to move through the giant metallic structure 3. Si / simple molecular substances do not conduct because there are no mobile charged particles / no delocalised electrons |
9701_w25_qp_22 Q2(a)(ii); 9701_s25_qp_24 Q1(b) |
| 7% | 3/44 | 3 | amphoteric oxides/hydroxides | 1. Al₂O₃/Al(OH)₃ are amphoteric: they react with both acids and bases to form salts两性氧化物方程 / Amphoteric oxide equations知识点归属Al₂O₃ + 6HCl → 2AlCl₃ + 3H₂O
Al₂O₃ + 2NaOH + 3H₂O → 2NaAl(OH)₄ or Al₂O₃ + 2NaOH → 2NaAlO₂ + H₂O 使用场景:1. use these to show amphoteric behaviour with acid and alkali
2. tetrahydroxoaluminate and aluminate forms have both appeared as accepted balanced answers |
9701_s20_qp_21 Q1(c)(ii); 9701_m20_qp_22 Q1(e)(i) |
| 2% | 1/44 | 1 | acidic/basic oxides | 1. metal oxides are basic 2. non-metal oxides are acidic 3. Al₂O₃/Al(OH)₃ are amphoteric. Give acid/base equations if asked |
9701_s21_qp_23 Q3(a)(i) |
10 Group 2
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 9% | 4/44 | 4 | Group 2 thermal stability | 1. down Group 2 the cation radius increases and charge density decreases 2. the cation polarises the carbonate/nitrate ion less 3. decomposition is less easy and thermal stability increases; a higher temperature is needed for decomposition |
9701_w21_qp_23 Q2(a)(iii); 9701_w21_qp_21 Q2(a)(iii); 9701_m21_qp_22 Q2(c)(i); +8 additional hits |
| 7% | 3/44 | 3 | basic oxides/hydroxides reaction | 1. Group 2 oxides/hydroxides neutralise acids to form salt and water 2. oxide ionic equation: MO + 2H⁺ → M²⁺ + H₂O 3. soluble oxides react with water to form hydroxides: MO + H₂O → M(OH)₂ |
9701_w20_qp_22 Q2(c) |
| 7% | 3/44 | 3 | hydroxide solubility trend | 1. Group 2 hydroxides become more soluble down the group | 9701_m23_qp_22 Q2(c); 9701_s22_qp_22 Q2(c)(iv) |
| 2% | 1/44 | 1 | sulfate solubility trend | 1. Group 2 sulfates become less soluble down the group 2. BaSO4 is insoluble |
9701_s23_qp_21 Q2(c)(ii); 9701_s23_qp_21 Q2(c)(i); 9701_m23_qp_22 Q2(c) |
11 Group 17
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 30% | 13/44 | 11 | chlorine disproportionation / water purification | 1. cold dilute NaOH: Cl₂ + 2NaOH → NaCl + NaClO + H₂O 2. hot concentrated NaOH: 3Cl₂ + 6NaOH → 5NaCl + NaClO₃ + 3H₂O 3. in water: Cl₂ + H₂O ⇌ HCl + HOCl 4. HOCl kills bacteria/microbes in water purification; ClO⁻ is the active species in alkaline bleach 5. chlorine disproportionates because Cl is both oxidised and reduced 氯水/歧化方程 / Chlorine equations知识点归属Cl₂ + H₂O ⇌ HCl + HOCl
Cl₂ + 2NaOH → NaCl + NaClO + H₂O 3Cl₂ + 6NaOH → 5NaCl + NaClO₃ + 3H₂O 使用场景:1. cold dilute alkali gives NaClO
2. hot concentrated alkali gives NaClO₃ 3. HOCl is the active species in water purification; ClO⁻ is used for alkaline bleach wording |
9701_m25_qp_22 Q3(b); 9701_s23_qp_22 Q2(d); 9701_s21_qp_23 Q1(c); +10 additional hits |
| 14% | 6/44 | 6 | AgNO₃ / NH₃ test | 1. acidify with dilute HNO₃, then add aqueous AgNO₃ 2. Cl⁻ gives a white precipitate, soluble in dilute NH₃ 3. Br⁻ gives a cream/off-white precipitate, partly/slightly soluble in concentrated NH₃ 4. I⁻ gives a yellow precipitate, insoluble in concentrated NH₃ |
9701_w22_qp_21 Q2(c); 9701_s22_qp_22 Q2(d); 9701_m20_qp_22 Q2(c)(i); +6 additional hits |
| 11% | 5/44 | 5 | halogen volatility | 1. more electrons down the group 2. stronger instantaneous dipole-induced dipole forces / van der Waals’ forces between molecules 3. more energy is required to overcome these forces 4. boiling point increases / volatility decreases |
9701_w21_qp_21 Q1(a)(iv); 9701_w20_qp_23 Q4(a)(i); 9701_w20_qp_21 Q4(a)(i); +3 additional hits |
| 9% | 4/44 | 4 | halide reducing power | 1. reducing power increases down the group 2. halide ion is oxidised / loses an electron more readily 3. outer electron is less strongly attracted because distance/shielding increases |
9701_s22_qp_22 Q3(d)(iii); 9701_m21_qp_22 Q2(c)(iii) |
| 7% | 3/44 | 3 | conc H₂SO₄ with halides | 1. with Cl⁻, H₂SO₄ acts as an acid only 2. with Br⁻/I⁻, H₂SO₄ is reduced and acts as an oxidising agent 3. Br₂/I₂ and sulfur-containing reduction products can form 卤离子氧化还原方程 / Halide redox equations跨章节引用displacement: X₂ + 2Y⁻ → 2X⁻ + Y₂
H₂SO₄ acid-base: NaX + H₂SO₄ → NaHSO₄ + HX bromide redox: 2Br⁻ + 2H⁺ + H₂SO₄ → Br₂ + 2H₂O + SO₂ iodide redox: 8HI + H₂SO₄ → 4I₂ + H₂S + 4H₂O 使用场景:1. choose X₂/Y⁻ from halogen reactivity
2. with concentrated H₂SO₄, Br⁻/I⁻ can reduce sulfuric acid 3. use oxidation numbers to justify redox |
9701_m22_qp_22 Q3(b)(ii); 9701_s21_qp_21 Q3(d); 9701_s21_qp_21 Q3(a)(i); 9701_m20_qp_22 Q2(c)(ii) |
| 5% | 2/44 | 2 | halogen oxidising power | 1. oxidising power decreases down the group 2. halogen atom gains an electron less readily 3. distance/shielding increases, so attraction for the incoming electron is weaker |
9701_s21_qp_23 Q1(b); 9701_m20_qp_22 Q2(b)(i) |
12 Nitrogen And Sulfur
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 25% | 11/44 | 15 | SO₂ acid rain | 1. SO₂ reacts with water to form H₂SO₃ 2. SO₂ / H₂SO₃ can be oxidised to H₂SO₄ 3. these acids cause acid rain |
9701_w21_qp_23 Q1(d)(ii); 9701_w21_qp_21 Q1(d)(ii); 9701_s20_qp_22 Q2(d); +3 additional hits |
| 20% | 9/44 | 11 | NOₓ / atmospheric pollution | 1. NOₓ forms when N₂ and O₂ react at high temperature in an internal combustion engine / lightning 2. NO/NO₂ reacts with oxygen and/or water/moist air to form HNO₂/HNO₃ and cause acid rain 3. NO/NO₂ can act as a homogeneous catalyst in SO₂ oxidation to SO₃ and is regenerated 4. NO₂/NOₓ reacts with unburned hydrocarbons/VOCs to form photochemical smog / PAN NOₓ / SO₂ 污染方程跨章节引用N₂ + O₂ → 2NO
NO + ½O₂ → NO₂ 3NO₂ + H₂O → 2HNO₃ + NO NO₂ + SO₂ → NO + SO₃ 2NO + O₂ → 2NO₂ SO₃ + H₂O → H₂SO₄ 使用场景:1. the two NO₂/SO₂ equations show NO₂ as a catalyst and regenerated
2. SO₃ + water can be written when MS allows equation form for sulfuric acid formation 3. NO forms from N₂/O₂ at high temperature |
9701_s24_qp_23 Q2(d)(i-ii); 9701_s23_qp_21 Q5(b)(i-iii); 9701_s22_qp_21 Q2(b)(i-ii); 9701_s20_qp_22 Q2(d); +5 additional hits |
| 16% | 7/44 | 7 | photochemical smog / PAN | 1. unburned/unburnt hydrocarbons / VOCs react or combine with NO and/or NO₂ 2. PAN / peroxyacetyl nitrate / peroxyacetylnitrate is formed 3. PAN is a component of photochemical smog |
9701_s25_qp_23 Q3(a)(ii); 9701_s24_qp_23 Q2(d)(ii); 9701_s22_qp_21 Q2(b)(i); 9701_m22_qp_22 Q2(d)(ii); +3 additional hits |
| 11% | 5/44 | 7 | ammonium ion / ammonium salts | 1. NH₄⁺ / ammonium ion is a Brønsted-Lowry acid / proton donor 2. NH₄⁺ + OH⁻ → NH₃ + H₂O; use alkali/NaOH and heat/warm to liberate NH₃ from ammonium salts 3. NH₃ accepts H⁺ using the lone pair on N; in NH₄⁺ there are four N-H bonds, one may be shown as coordinate when the formation is asked 氨/铵根方程 / Ammonia equations知识点归属NH₃ + H₂O ⇌ NH₄⁺ + OH⁻
NH₄⁺ + OH⁻ → NH₃ + H₂O 使用场景:1. reversible equation shows weak base behaviour
2. ionic equation is used for ammonium salt + alkali test |
9701_s24_qp_23 Q4(a-b); 9701_w22_qp_22 Q1(c)(ii); 9701_s22_qp_21 Q2(c); +2 additional hits |
| 9% | 4/44 | 4 | N₂ lack of reactivity | 1. N₂ molecules have a strong N≡N triple covalent bond / high N≡N bond enthalpy 2. high activation energy / much energy is needed to break the N≡N bond 3. N₂ molecules are non-polar 4. in comparison questions, P≡P is much weaker, so P₂ is more reactive than N₂ |
9701_w24_qp_22 Q3(f)(ii); 9701_s24_qp_23 Q1(a)(i); 9701_m24_qp_22 Q3(a); 9701_m21_qp_22 Q3(a)(ii) |
| 9% | 4/44 | 4 | ammonia / Bronsted base | 1. NH₃ is a Bronsted-Lowry base/proton acceptor 2. the lone pair on N accepts H⁺ |
9701_w24_qp_22 Q3(b)(iii) |
| 2% | 1/44 | 1 | homogeneous catalyst | 1. homogeneous catalyst is in the same phase/state as the reactants 2. it increases rate / lowers Ea 3. it is regenerated / chemically unchanged overall |
9701_s20_qp_21 Q3(e)(i) |
13 Introduction To AS Organic Chemistry
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 43% | 19/44 | 14 | σ/π bond counting and hybridisation in organic molecules | 1. count every single bond as one σ bond 2. each C=C contains one σ bond and one π bond; each C≡N/C≡C contains one σ bond and two π bonds 3. π bonds are formed by sideways/lateral overlap of p orbitals 4. sp² centres are trigonal planar around C=C; sp³ centres are tetrahedral around saturated carbon |
9701_s24_qp_21 Q4(b)(i-ii); 9701_s23_qp_22 Q3(c); 9701_s20_qp_23 Q4(d)(ii); +16 additional hits |
| 18% | 8/44 | 8 | structural isomerism | 1. same molecular formula but different structural formulae | 9701_w22_qp_22 Q3(a)(ii); 9701_w22_qp_21 Q3(d)(iv); 9701_s21_qp_23 Q5(d); +6 additional hits |
| 16% | 7/44 | 7 | stereoisomerism | 1. same structural formula but different spatial arrangement of atoms/groups | 9701_w22_qp_23 Q5(a)(iii); 9701_w22_qp_21 Q5(a)(iii); 9701_w21_qp_22 Q3(e)(iv); +8 additional hits |
| 11% | 5/44 | 5 | geometrical isomerism | 1. restricted rotation about C=C 2. each carbon atom in the C=C has two different groups attached |
9701_s20_qp_23 Q5(a)(iii); 9701_s20_qp_22 Q2(c)(ii); 9701_m20_qp_22 Q3(d)(i); +7 additional hits |
| 9% | 4/44 | 4 | chiral centre / optical isomerism | 1. chiral carbon/centre is bonded to four different groups 2. enantiomers are non-superimposable mirror images |
9701_s21_qp_23 Q5(b)(i); 9701_s20_qp_23 Q3(b)(ii); 9701_m20_qp_22 Q3(a)(iv); +8 additional hits |
| 2% | 1/44 | 1 | functional group | 1. for identification questions, name the functional group exactly 2. definition: atom or group of atoms responsible for the characteristic reactions of the compound |
9701_s21_qp_22 Q5(b)(iv); 9701_s21_qp_22 Q4(d)(ii); 9701_s20_qp_23 Q5(b)(i); +4 additional hits |
14 Hydrocarbons
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 27% | 12/44 | 12 | major product / carbocation stability | 1. major product forms via the more stable carbocation/intermediate 2. more alkyl groups give greater positive inductive effect/electron donation 3. positive charge is spread/stabilised more |
9701_s20_qp_21 Q5(d)(ii); 9701_s20_qp_21 Q5(d)(i); 9701_m20_qp_22 Q3(c)(iii); +30 additional hits |
| 23% | 10/44 | 13 | alkene test | 1. bromine water / Br₂(aq) is decolourised from orange/brown to colourless | 9701_s22_qp_22 Q6(a); 9701_s21_qp_21 Q5(c)(ii); 9701_s21_qp_21 Q4(b); +7 additional hits |
| 18% | 8/44 | 25 | radical substitution mechanism | 1. initiation: homolytic fission in UV light forms halogen radicals 2. propagation 1: a halogen radical removes H from an alkane to form an alkyl radical and HCl/HBr 3. propagation 2: the alkyl radical reacts with Cl2/Br2 to form a halogenoalkane and regenerate a halogen radical 4. termination: two radicals combine to form a stable molecule 自由基取代链式方程 / Radical substitution equations知识点归属initiation: Cl₂ / Br₂ → 2Cl· / 2Br·
propagation 1: RH + Cl· → R· + HCl propagation 2: R· + Cl₂ → RCl + Cl· termination: R· + Cl· → RCl or R· + R· → R-R 使用场景:1. radical dot must be on the radical species
2. initiation needs homolytic fission/UV context if asked 3. propagation regenerates a halogen radical |
9701_w24_qp_21 Q2(b)(iii); 9701_m21_qp_22 Q2(g)(i); 9701_s20_qp_23 Q4(b)(ii); +22 additional hits |
| 7% | 3/44 | 3 | addition reaction definition | 1. two or more molecules combine to form a single product 2. no atoms are lost |
9701_w20_qp_23 Q4(b)(ii); 9701_w20_qp_21 Q4(b)(ii); 9701_s20_qp_22 Q5(a); +3 additional hits |
| 7% | 3/44 | 3 | free radical definition | 1. species with one or more unpaired electrons | 9701_w24_qp_23 Q2(b)(i); 9701_w24_qp_21 Q2(b)(i); 9701_w23_qp_22 Q2(e)(i) |
| 5% | 2/44 | 2 | incomplete combustion pollutants | 1. incomplete combustion can produce C/soot, CO and unburnt/unburned hydrocarbons, with less/no CO₂ 2. CO is toxic/poisonous/harmful to health 3. unburnt/unburned hydrocarbons can catalyse/contribute to photochemical smog |
9701_s23_qp_21 Q5(a)(ii); 9701_s21_qp_21 Q5(e)(ii) |
| 5% | 2/44 | 3 | homolytic fission / radical formation | 1. ultraviolet light supplies energy for homolytic fission 2. the covalent bond breaks evenly so each atom/species receives one electron 3. radicals with unpaired electrons are formed |
9701_s22_qp_22 Q4(b)(ii); 9701_s20_qp_21 Q5(b)(ii); 9701_s20_qp_21 Q5(b)(i) |
| 2% | 1/44 | 1 | alkane combustion / cracking | 1. complete combustion forms CO₂ and H₂O 2. cracking uses heat and a catalyst such as Al₂O₃/SiO₂ to form a shorter alkane and an alkene |
9701_s20_qp_23 Q1(b)(i); 9701_s20_qp_21 Q6(f)(i); 9701_s20_qp_21 Q4(c); +20 additional hits |
15 Halogen Compounds
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 9% | 4/44 | 4 | nucleophilic substitution | 1. curly arrow from lone pair/electron pair on the nucleophile to the electron-deficient carbon 2. curly arrow from the C-X bond to X as the C-X bond breaks 3. halide ion leaves 卤代烷取代/消除方程知识点归属substitution: RCl + NaOH(aq) → ROH + NaCl
elimination: RCl + NaOH(ethanol) → alkene + NaCl + H₂O 使用场景:1. aqueous hydroxide gives alcohol
2. ethanolic hydroxide with heat/reflux gives alkene 3. adapt the carbon skeleton from the question |
9701_s21_qp_22 Q3(c)(i); 9701_w20_qp_23 Q4(c)(ii); 9701_w20_qp_21 Q4(c)(ii); +2 additional hits |
| 5% | 2/44 | 2 | C-X bond strength and hydrolysis rate | 1. C-I has lower bond enthalpy / is weaker than C-Cl 2. I is larger, so the shared pair is less strongly attracted to the iodine nucleus 3. less energy is needed to break C-I / form the carbocation, so Ea is lower 4. hydrolysis/substitution is faster |
9701_s22_qp_22 Q5(b); 9701_s21_qp_22 Q3(d) |
16 Hydroxy Compounds
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 5% | 2/44 | 2 | iodoform reaction | 1. a CH₃CO- or CH₃CH(OH)- group is present 2. alkaline aqueous iodine / I₂ and NaOH is used 3. yellow precipitate of CHI₃ forms 碘仿/卤仿方程 / Iodoform equations知识点归属CH₃COCH₃ + 3I₂ + 4OH⁻ → CH₃COO⁻ + 3H₂O + 3I⁻ + CHI₃
CH₃COCH₃ + 3NaClO → CHCl₃ + CH₃COONa + 2NaOH 使用场景:1. CHI₃ / CHCl₃ product is often a named-product mark
2. balancing may be a separate mark 3. use for methyl ketone / suitable secondary alcohol context |
9701_s22_qp_21 Q4(c)(ii); 9701_w20_qp_23 Q4(c)(vi); 9701_w20_qp_21 Q4(c)(vi); +2 additional hits |
| 2% | 1/44 | 1 | Na with alcohol | 1. alcohol reacts with Na to give effervescence of H₂ and sodium alkoxide 2. Na is oxidised from 0 to +1 |
9701_s20_qp_22 Q4(c)(ii); 9701_s20_qp_22 Q1(e)(ii); 9701_s20_qp_21 Q4(c); +59 additional hits |
| 2% | 1/44 | 1 | alcohol oxidation conditions/products | 1. primary alcohol + acidified K₂Cr₂O₇/KMnO₄: distil to aldehyde 2. primary alcohol + acidified K₂Cr₂O₇/KMnO₄: reflux to carboxylic acid 3. secondary alcohol refluxes to ketone 4. tertiary alcohol shows no oxidation |
9701_s21_qp_23 Q5(b)(ii); 9701_m21_qp_22 Q4(c)(ii) |
| 2% | 1/44 | 1 | tertiary alcohol oxidation | 1. tertiary alcohol is not oxidised because there is no H attached to the carbon bearing the -OH group | 9701_s21_qp_23 Q5(b)(ii) |
17 Carbonyl Compounds
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 23% | 10/44 | 10 | 2,4-DNPH test | 1. 2,4-DNPH gives a red/orange/yellow precipitate with carbonyl compounds (aldehydes/ketones) | 9701_s21_qp_23 Q4(b); 9701_w20_qp_22 Q4(c); 9701_s20_qp_21 Q6(d); +10 additional hits |
| 20% | 9/44 | 9 | Tollens / Fehling distinction | 1. aldehydes reduce Tollens’ reagent to a silver mirror/grey precipitate 2. aldehydes reduce Fehling’s solution to a brick-red precipitate 3. ketones show no visible change |
9701_m22_qp_22 Q4(a)(i); 9701_m21_qp_22 Q4(b); 9701_w20_qp_22 Q4(a)(iv); +6 additional hits |
| 9% | 4/44 | 4 | HCN nucleophilic addition mechanism | 1. use HCN with a trace amount of NaCN/KCN to provide CN⁻; heat is not required unless specified by the question 2. curly arrow from the lone pair on CN⁻ to the δ⁺ carbonyl carbon 3. curly arrow from the C=O π bond to O 4. O⁻/alkoxide is protonated by HCN/H⁺ to form the hydroxynitrile and regenerate CN⁻ if shown |
9701_w20_qp_23 Q3(c)(iii); 9701_w20_qp_21 Q3(c)(iii); 9701_s20_qp_22 Q5(b)(i); +8 additional hits |
| 7% | 3/44 | 3 | carbonyl reduction | 1. NaBH₄ or LiAlH₄ reduces aldehydes to primary alcohols and ketones to secondary alcohols | 9701_w20_qp_23 Q3(c)(ii); 9701_w20_qp_22 Q3(c)(i); 9701_w20_qp_21 Q3(c)(ii); +5 additional hits |
| 2% | 1/44 | 1 | aldehyde oxidation | 1. aldehydes oxidise to carboxylic acids with acidified K₂Cr₂O₇ or acidified KMnO₄ 2. acidified K₂Cr₂O₇ changes from orange to green; acidified KMnO₄ changes from purple to colourless / pale pink 3. Tollens’ and Fehling’s reagents are alkaline tests; aldehydes give silver mirror / brick-red precipitate, ketones show no visible change 4. ketones are not oxidised under these conditions |
9701_s21_qp_22 Q4(d)(ii) |
18 Carboxylic Acids And Derivatives
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 14% | 6/44 | 6 | carboxylic acid + carbonate/metal | 1. carbonate/hydrogencarbonate gives effervescence of CO₂ 2. CO₂ turns limewater milky if tested 3. reactive metal gives effervescence of H₂ and a salt |
9701_m25_qp_22 Q2(d)(i); 9701_s23_qp_23 Q5(d); 9701_s22_qp_21 Q4(b)(i) |
| 11% | 5/44 | 5 | esterification | 1. carboxylic acid + alcohol with concentrated H₂SO₄ catalyst and heat/reflux forms ester + water酯化/羧酸反应方程知识点归属esterification: RCOOH + R′OH → RCOOR′ + H₂O
neutralisation: RCOOH + NaOH → RCOONa + H₂O carbonate: RCOOH + carbonate → carboxylate salt + CO₂ + H₂O 使用场景:1. ester formula must connect acyl O correctly
2. carbonate reaction is useful as a carboxylic acid test template 3. adapt salt stoichiometry to mono-/di-carboxylic acids |
9701_w21_qp_22 Q3(e)(v); 9701_w20_qp_22 Q4(a)(v); 9701_m20_qp_22 Q3(a)(v) |
| 2% | 1/44 | 1 | ester hydrolysis | 1. dilute acid + heat/reflux gives carboxylic acid + alcohol 2. NaOH(aq) + heat gives carboxylate + alcohol 3. acidification gives the carboxylic acid |
9701_s21_qp_23 Q4(c) |
19 Nitrogen Compounds
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 23% | 10/44 | 10 | nitrile hydrolysis to carboxylic acid / carboxylate | 1. acid hydrolysis: RCN + 2H₂O + HCl/H⁺ → RCOOH + NH₄Cl/NH₄⁺ 2. alkaline hydrolysis: RCN + 2H₂O + NaOH → RCO₂Na + NH₄OH/NH₃; acidification gives RCOOH if required 3. heat/reflux with dilute acid or aqueous alkali 4. the nitrile carbon becomes the carboxyl carbon, so the carbon chain length does not change during hydrolysis 腈水解/还原方程 / Nitrile equations知识点归属acid hydrolysis: RCN + 2H₂O + H⁺ → RCOOH + NH₄⁺
alkaline hydrolysis: RCN + NaOH + H₂O → RCO₂Na + NH₃ reduction: RCN + 2H₂ → RCH₂NH₂ 使用场景:1. acid hydrolysis gives carboxylic acid + ammonium
2. alkaline hydrolysis gives carboxylate salt + ammonia 3. the nitrile carbon becomes the carboxyl/amine carbon |
9701_s25_qp_24 Q4(d)(i); 9701_s22_qp_22 Q5(c)(ii); 9701_w20_qp_21 Q3(c)(iv); +7 additional hits |
| 9% | 4/44 | 4 | hydroxynitrile preparation | 1. aldehyde/ketone + HCN with a trace amount of KCN/NaCN catalyst 2. CN⁻ attacks the δ⁺ carbonyl carbon and nucleophilic addition forms a hydroxynitrile 3. the new C-C bond adds one carbon to the carbon chain |
9701_m25_qp_22 Q4(b)(ii) |
20 Polymerisation
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 7% | 3/44 | 3 | repeat unit drawing | 1. one repeat unit with correct carbon backbone and continuation/dangling bonds | 9701_w22_qp_23 Q4(d)(ii); 9701_w22_qp_22 Q3(b)(ii); 9701_w22_qp_21 Q4(d)(ii) |
| 2% | 1/44 | 1 | addition polymerisation definition | 1. alkene monomers join as C=C bonds open 2. no small molecule is eliminated |
9701_m21_qp_22 Q3(d)(i) |
| 2% | 1/44 | 1 | polymer disposal | 1. addition polymers are often non-biodegradable 2. combustion of chlorine-containing polymers can produce HCl/toxic gases |
9701_s22_qp_22 Q4(c)(ii) |
21 Organic Synthesis
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 41% | 18/44 | 18 | oxidation/reduction route | 1. choose reagent and conditions from the functional group 2. acidified K₂Cr₂O₇/KMnO₄ with distil/reflux for alcohols/aldehydes 3. NaBH₄ or LiAlH₄ for carbonyl reduction 氧化/还原通式 / Redox templates跨章节引用oxidation: RCH₂OH + 2[O] → RCO₂H + H₂O; R₂CHOH + [O] → R₂CO + H₂O
reduction: RCHO + 2[H] → RCH₂OH; R₂CO + 2[H] → R₂CHOH; RCO₂H + 4[H] → RCH₂OH + H₂O 使用场景:1. primary alcohol to acid needs 2[O]
2. secondary alcohol to ketone needs [O] 3. carboxylic acid reduction needs 4[H] and forms water |
9701_w20_qp_21 Q3(c)(i); 9701_s20_qp_22 Q5(a); 9701_m20_qp_22 Q3(a)(i); +22 additional hits |
| 5% | 2/44 | 2 | multi-step route reagents/conditions | 1. state reagent and condition for every step, and name the reaction type when asked 2. each is often a separate mark |
9701_s20_qp_22 Q5(a) |
22 Analytical Techniques
| Frequency | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|
| 36% | 16/44 | 17 | IR absorption identification | 1. quote the absorption range in cm⁻¹ 2. state the bond responsible 3. state the functional group responsible 4. use presence/absence of O-H, C=O, C≡N etc. as evidence |
9701_w20_qp_21 Q3(c)(v); 9701_s20_qp_21 Q6(e); 9701_m20_qp_22 Q3(d)(iii); +17 additional hits |
| 11% | 5/44 | 5 | M+1 carbon count | 1. use the abundance of the [M+1]⁺ peak 2. number of carbon atoms = abundance of [M+1]⁺ peak / (1.1 × abundance of M⁺ peak) |
9701_w25_qp_21 Q4(c)(iii); 9701_s23_qp_23 Q5(c); 9701_s23_qp_21 Q4(b)(iii); +1 additional hits |
| 7% | 3/44 | 3 | molecular ion peak | 1. molecular ion peak M⁺ gives the Mr / molecular mass 2. the molecular ion is the molecule with one electron removed and no bond broken |
9701_s22_qp_23 Q5(b)(ii); 9701_s22_qp_22 Q6(c)(i) |
| 16% | 7/44 | 7 | mass spectrometry fragmentation | 1. fragment ions are formed when molecular ions break into smaller positive ions/radicals 2. m/z of a fragment helps identify part of the molecule 3. common hydrocarbon fragments include CH₃⁺ at m/z 15, C₂H₅⁺ at m/z 29 and C₃H₇⁺ at m/z 43, if supported by the question data |
9701_w25_qp_23; 9701_w25_qp_21; 9701_s25_qp_24; 9701_s24_qp_22; 9701_s22_qp_23; +2 additional hits |
| 5% | 2/44 | 2 | relative atomic mass from isotopes | 1. relative atomic mass = sum of isotope mass × fractional abundance 2. divide percentage abundances by 100 |
9701_s22_qp_21 Q1(e)(ii); 9701_s20_qp_23 Q2(b); 9701_s20_qp_21 Q1(a)(ii); +5 additional hits |
Appendix: Syllabus / Low-Frequency Supporting Points
以下条目保留 syllabus/textbook/core review points,或属于具体路线/基础技能而未放入主表高频排序。Freq / Papers / MS hits 按当前 2020-2025 P2 MS 来源校正;no 2020-2025 P2 MS hit 表示本轮统计范围内没有 recent P2 MS 来源。
| Section | Freq | Papers | MS hits | Point | Full-mark wording | Sources |
|---|---|---|---|---|---|---|
| 1 Atomic Structure / Electrons In Atoms | 2% | 1/44 | 1 | first ionisation energy definition | 1. energy required when one electron is removed 2. from each atom in one mole of gaseous atoms |
9701_s22_qp_23 Q1(a) |
| 1 Atomic Structure / Electrons In Atoms | 0% | 0/44 | 0 | ground state definition | 1. lowest energy arrangement of electrons | no 2020-2025 P2 MS hit |
| 2 Atoms, Molecules And Stoichiometry | 0% | 0/44 | 0 | limiting reagent | 1. reagent used up first according to the stoichiometric ratio | no 2020-2025 P2 MS hit |
| 4 States Of Matter | 0% | 0/44 | 0 | vapour pressure definition | 1. pressure exerted by vapour in equilibrium with liquid at a stated temperature in a closed system | no 2020-2025 P2 MS hit |
| 5 Chemical Energetics | 0% | 0/44 | 0 | Hess law cycle/signs | 1. reverse arrows change the sign of ΔH 2. multiply ΔH by coefficients when equations are multiplied 3. combine steps to obtain the target ΔH |
no 2020-2025 P2 MS hit |
| 5 Chemical Energetics | 2% | 1/44 | 1 | enthalpy change of neutralisation | 1. enthalpy/energy change when one mole of water is formed 2. from an aqueous acid and an aqueous alkali/base |
9701_s24_qp_21 Q2(c)(iii) |
| 8 Reaction Kinetics | 0% | 0/44 | 0 | surface area effect | 1. larger surface area exposes more particles, increasing the frequency of successful/effective collisions | no 2020-2025 P2 MS hit |
| 10 Group 2 | 0% | 0/44 | 0 | Group 2 reactivity trend | 1. down Group 2, shielding and distance of the outer electron from the nucleus increase 2. nuclear attraction for the outer electron decreases / first and second ionisation energies decrease 3. electrons are lost more readily, so reactivity increases |
no 2020-2025 P2 MS hit |
| 11 Group 17 | 5% | 2/44 | 2 | hydrogen halide thermal stability | 1. H-X covalent bond strength decreases down the group 2. X atom is larger / H-X bond is longer, so there is weaker attraction between the nucleus and the shared pair 3. less energy is needed to break the bond, so thermal stability decreases |
9701_w24_qp_23 Q2(a)(iv); 9701_w24_qp_21 Q2(a)(iv) |
| 12 Nitrogen And Sulfur | 20% | 9/44 | 9 | Contact/Haber equilibrium conditions | 1. link temperature to exothermic/endothermic direction and equilibrium shift/yield 2. link pressure to the side with fewer moles of gas 3. chosen conditions are a compromise between yield, rate and cost |
9701_w21_qp_23 Q1(d)(i); 9701_w21_qp_21 Q1(d)(i); 9701_s20_qp_21 Q3(e)(ii); +6 additional hits |
| 13 Introduction To AS Organic Chemistry | 0% | 0/44 | 0 | hydrocarbon definition | 1. compound containing carbon and hydrogen only | no 2020-2025 P2 MS hit |
| 13 Introduction To AS Organic Chemistry | 0% | 0/44 | 0 | homologous series definition | 1. same functional group and same general formula 2. successive members differ by CH₂ and have similar chemical properties |
no 2020-2025 P2 MS hit |
| 15 Halogen Compounds | 16% | 7/44 | 7 | KCN chain extension | 1. reagent is KCN/NaCN 2. condition is ethanol/alcohol and heat under reflux 3. nucleophilic substitution forms a nitrile and increases the carbon chain by one |
9701_s23_qp_22 Q3(a)(i); 9701_s21_qp_23 Q4(c); 9701_s21_qp_22 Q5(b)(iii); +4 additional hits |
| 15 Halogen Compounds | 5% | 2/44 | 2 | NH₃ to amine | 1. reagent is excess NH₃/ammonia 2. condition is ethanol/alcohol with heat under pressure / sealed tube 3. nucleophilic substitution forms a primary amine |
9701_s24_qp_23 Q4(e)(iii); 9701_m21_qp_22 Q3(d)(iii) |
| 15 Halogen Compounds | 5% | 2/44 | 2 | aqueous vs ethanolic NaOH | 1. hot aqueous NaOH gives nucleophilic substitution to an alcohol 2. NaOH/KOH in ethanol/alcohol and heat under reflux gives elimination to an alkene |
9701_s22_qp_22 Q5(a); 9701_m22_qp_22 Q4(b)(iv) |
| 15 Halogen Compounds | 2% | 1/44 | 1 | tertiary halogenoalkane definition | 1. the carbon atom bonded to the halogen atom is attached directly to three alkyl groups / three carbon atoms | 9701_s20_qp_21 Q5(c)(ii) |
| 16 Hydroxy Compounds | 0% | 0/44 | 0 | alcohol acidity | 1. alcohols are weaker acids than water because alkyl groups donate electron density, making RO- less stable | no 2020-2025 P2 MS hit |
| 18 Carboxylic Acids And Derivatives | 9% | 4/44 | 4 | carboxylic acid acidity | 1. carboxylate ion is stabilised 2. negative charge is delocalised over the two oxygen atoms 3. the conjugate base is more stable, so the acid donates H⁺ more readily |
9701_w21_qp_21 Q4(b); 9701_w20_qp_23 Q3(c)(ii); 9701_w20_qp_21 Q3(c)(ii); +1 additional hits |
| 19 Nitrogen Compounds | 11% | 5/44 | 5 | amine basicity | 1. lone pair on N accepts H⁺ / a proton 2. the lone pair forms a dative bond to H⁺ 3. alkyl groups donate electron density to N if comparing basicity |
9701_w24_qp_21 Q4(a)(i); 9701_s23_qp_23 Q6(b)(ii); 9701_m23_qp_22 Q3(b)(i); +2 additional hits |
| 19 Nitrogen Compounds | 16% | 7/44 | 7 | nitrile preparation | 1. reagent is KCN/NaCN 2. condition is ethanol/alcohol and heat under reflux 3. nucleophilic substitution of a halogenoalkane forms a nitrile |
9701_s23_qp_22 Q3(a)(i); 9701_s21_qp_23 Q4(c); 9701_s21_qp_22 Q5(b)(iii); +4 additional hits |
| 19 Nitrogen Compounds | 2% | 1/44 | 1 | primary amine preparation | 1. reagent is excess NH₃/ammonia 2. condition is ethanol/alcohol with heat under pressure / sealed tube 3. nucleophilic substitution forms a primary amine |
9701_s24_qp_23 Q4(c)(ii) |
| 20 Polymerisation | 0% | 0/44 | 0 | monomer from polymer | 1. identify the repeat unit 2. then put C=C back between the two backbone carbon atoms to get the alkene monomer |
no 2020-2025 P2 MS hit |
| 21 Organic Synthesis | 0% | 0/44 | 0 | functional group identification | 1. use syllabus tests/reactions to identify functional groups before choosing a route 2. concrete functional-group naming, IR and chemical-test questions are counted in their own rows |
no 2020-2025 P2 MS hit |
| 21 Organic Synthesis | 0% | 0/44 | 0 | by-products | 1. analyse possible inorganic/organic by-products from substitution, elimination or hydrolysis | no 2020-2025 P2 MS hit |
| 22 Analytical Techniques | 2% | 1/44 | 1 | Cl/Br M+2 pattern | 1. one Cl gives M:M+2 about 3:1 because M+2 contains ³⁷Cl instead of ³⁵Cl 2. one Br gives M:M+2 about 1:1 because ⁷⁹Br and ⁸¹Br have similar abundance |
9701_s22_qp_23 Q5(b)(ii) |
| 22 Analytical Techniques | 5% | 2/44 | 2 | fragment ion formula | 1. assign each m/e or m/z peak to a possible fragment ion 2. include the positive charge on the fragment ion 3. use isotope composition where M/M+2 peaks are involved |
9701_s25_qp_21 Q2(b)(i); 9701_s23_qp_21 Q3(f) |