| 64% |
28/44 |
14-17 |
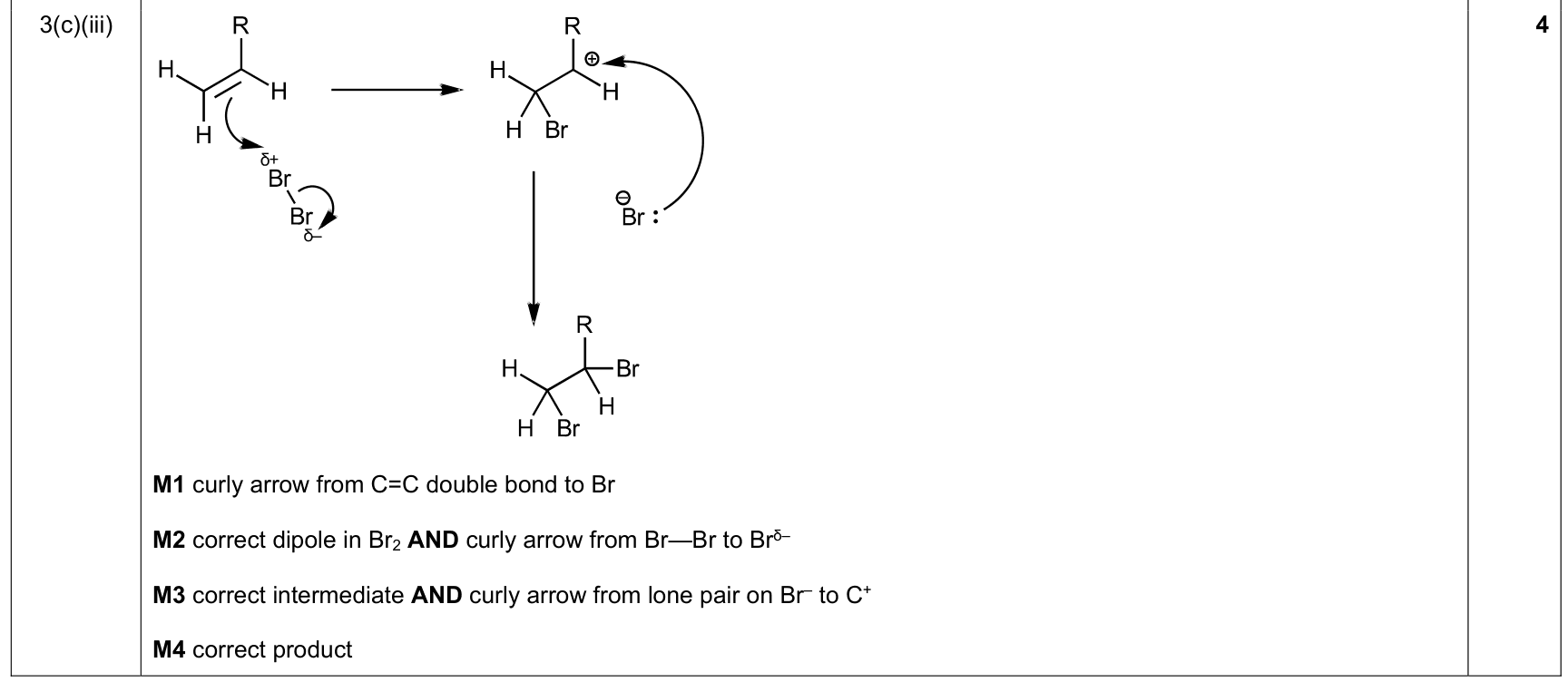
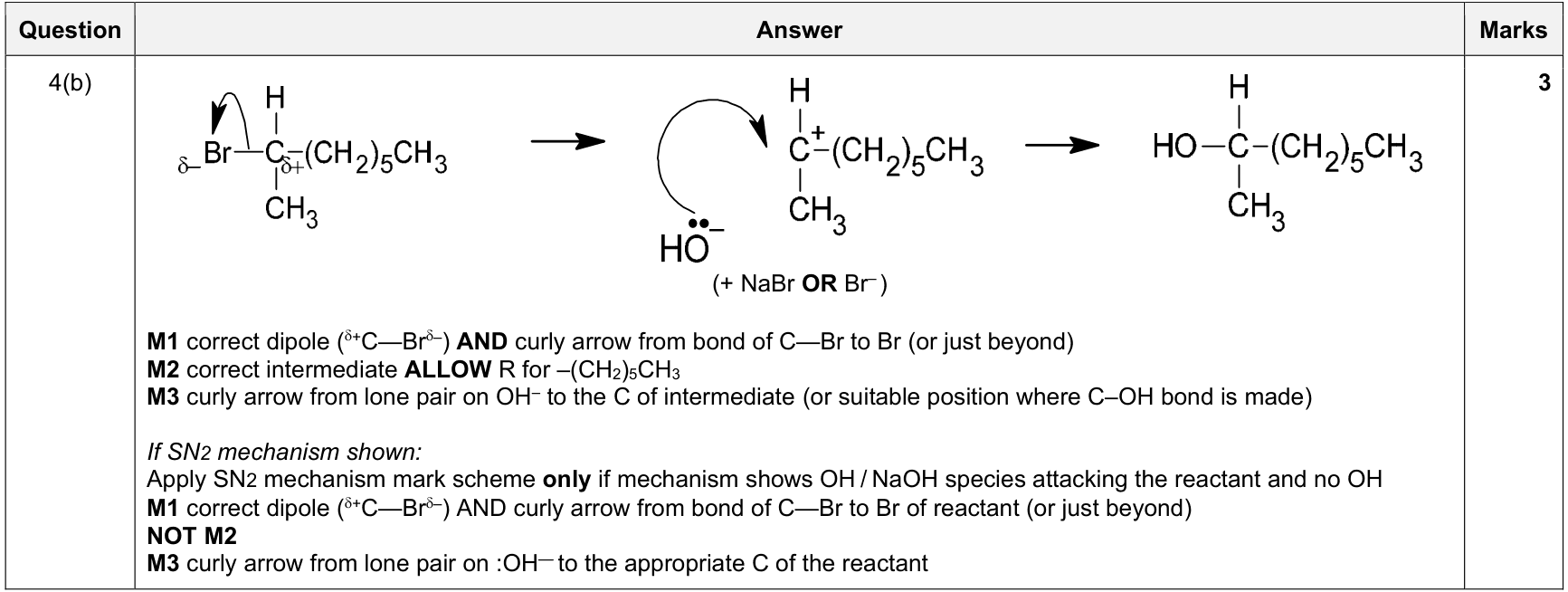
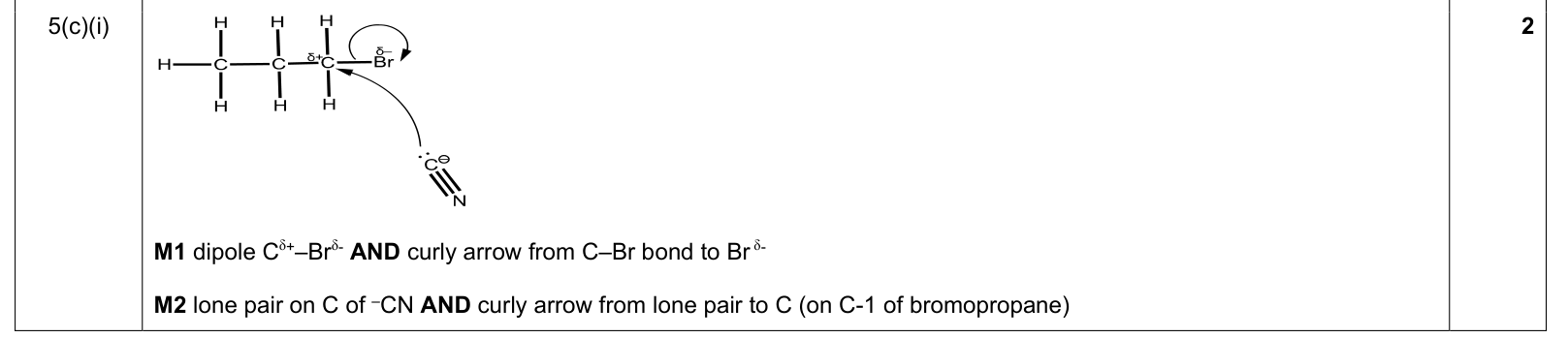
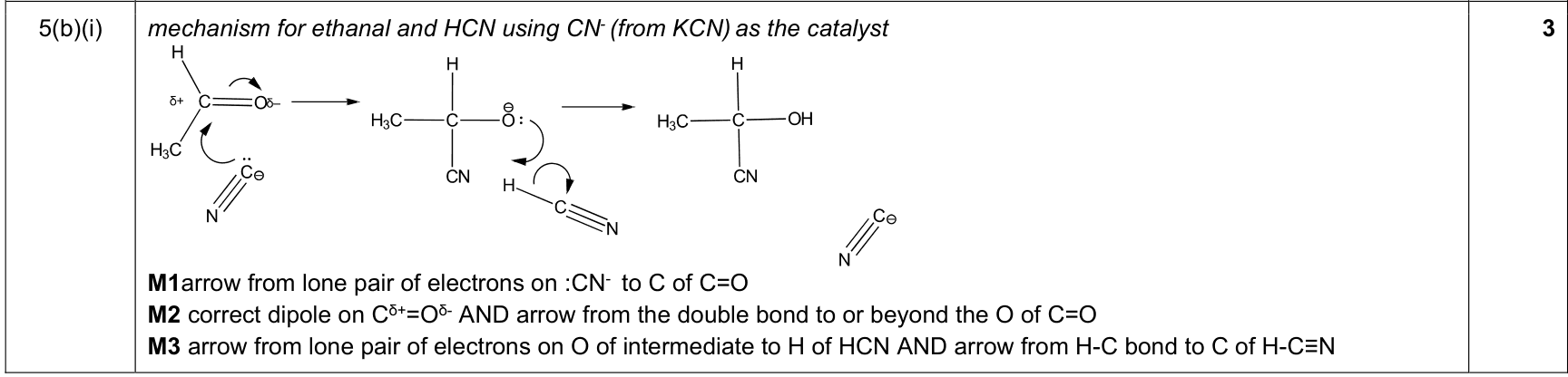
mechanism curly-arrow rule |
1. curly arrows start at a lone pair/electron pair/bond/negative charge
2. arrow points to the atom or bond receiving the electron pair
3. for bond breaking, arrow goes from the bond to the atom receiving the electrons |
9701_w25_qp_24 Q3(b)(ii); 9701_s25_qp_22 Q3(a)(ii); 9701_m25_qp_22 Q4(b)(i); +25 additional hits |
| 59% |
26/44 |
3 |
bonding diagrams: metallic / dot-and-cross / coordinate |
1. for dot-and-cross diagrams, show outer-shell electrons only and use different symbols for electrons from different atoms
2. covalent bonds are shared pairs; coordinate/dative bonds have both electrons in the shared pair donated by one atom/species
3. metallic bonding diagrams need a lattice/regular arrangement of positive ions/cations and delocalised electrons
4. for NH₄Cl, show ionic attraction between NH₄⁺ and Cl⁻, four N-H covalent bonds in NH₄⁺, and one N→H coordinate bond / one N-H bonding pair from N |
9701_s24_qp_23 Q4(a); 9701_w20_qp_21 Q3(a)(i); 9701_s20_qp_22 Q1(b); +23 additional hits |
| 50% |
22/44 |
3 |
simple molecular boiling/melting |
1. greater number of electrons in the molecules
2. stronger instantaneous dipole-induced dipole forces / van der Waals’ forces between molecules
3. more energy is needed to overcome the intermolecular forces |
9701_w20_qp_23 Q4(a)(iii); 9701_w20_qp_21 Q4(a)(iii); 9701_s20_qp_21 Q1(b)(i); +21 additional hits |
| 43% |
19/44 |
3 / 13 |
σ and π bonds / orbital overlap / hybridisation |
1. a σ bond is formed by direct/head-on/end-on overlap of orbitals between the bonding atoms
2. a π bond is formed by sideways/lateral overlap of adjacent p orbitals
3. single bond = 1σ; double bond = 1σ + 1π; triple bond = 1σ + 2π
4. common hybridisation: sp gives linear/triple-bond centres; sp² gives trigonal planar/C=C centres; sp³ gives tetrahedral/saturated carbon or ammonium centres |
9701_s24_qp_23 Q1(a)(ii); 9701_s24_qp_21 Q4(b)(i-ii); 9701_s22_qp_21 Q2(a); +16 additional hits |
| 41% |
18/44 |
21 |
oxidation/reduction route |
1. choose reagent and conditions from the functional group
2. acidified K₂Cr₂O₇/KMnO₄ with distil/reflux for alcohols/aldehydes
3. NaBH₄ or LiAlH₄ for carbonyl reduction |
9701_w20_qp_21 Q3(c)(i); 9701_s20_qp_22 Q5(a); 9701_m20_qp_22 Q3(a)(i); +22 additional hits |
| 36% |
16/44 |
all |
equations / state symbols |
1. use correct formulae
2. balance the equation
3. include charges where needed
4. include state symbols when the question asks for them |
9701_s20_qp_22 Q1(e)(ii); 9701_s20_qp_21 Q2(a)(i); 9701_s20_qp_21 Q1(c)(i); +20 additional hits |
| 36% |
16/44 |
1 |
IE trend explanation |
1. compare nuclear charge/proton number
2. compare shielding and distance from the nucleus
3. link these to nuclear attraction for the outer electron |
9701_w20_qp_22 Q1(b)(iv); 9701_w20_qp_21 Q1(c); 9701_w20_qp_21 Q1(b); +24 additional hits |
| 36% |
16/44 |
22 |
IR absorption identification |
1. quote the absorption range in cm⁻¹
2. state the bond responsible
3. state the functional group responsible
4. use presence/absence of O-H, C=O, C≡N etc. as evidence |
9701_w20_qp_21 Q3(c)(v); 9701_s20_qp_21 Q6(e); 9701_m20_qp_22 Q3(d)(iii); +17 additional hits |
| 32% |
14/44 |
9 |
Period 3 melting point structure |
1. identify the structure: simple molecular / giant metallic / giant covalent as appropriate
2. simple molecular substances: only weak intermolecular / van der Waals’ forces are overcome, so less energy is needed
3. giant metallic/covalent substances: many strong metallic/covalent bonds or attractions must be overcome/broken, so more energy is needed |
9701_w21_qp_23 Q3(a)(i); 9701_w21_qp_21 Q3(a)(i); 9701_w20_qp_22 Q2(f); +12 additional hits |
| 32% |
14/44 |
6 |
reducing agent |
1. donates electrons and is oxidised |
9701_w20_qp_22 Q4(b)(iii); 9701_w20_qp_21 Q3(c)(ii); 9701_m20_qp_22 Q2(c)(ii); +14 additional hits |
| 32% |
14/44 |
3 |
metallic bonding / conductivity |
1. giant metallic lattice / regular arrangement of positive ions/cations surrounded by delocalised electrons
2. many strong electrostatic attractions between cations and delocalised electrons / strong metallic bonds need more energy to overcome, so melting point is high
3. delocalised electrons are free to move through the structure and carry charge |
9701_w24_qp_21 Q3(a)(iii); 9701_s22_qp_23 Q1(f)(ii); 9701_s22_qp_22 Q1(a); +5 additional hits |
| 30% |
13/44 |
11 |
chlorine disproportionation / water purification |
1. cold dilute NaOH: Cl₂ + 2NaOH → NaCl + NaClO + H₂O
2. hot concentrated NaOH: 3Cl₂ + 6NaOH → 5NaCl + NaClO₃ + 3H₂O
3. in water: Cl₂ + H₂O ⇌ HCl + HOCl
4. HOCl / ClO⁻ kills bacteria/microbes; this is the active species in water purification
5. chlorine disproportionates because Cl is both oxidised and reduced |
9701_m25_qp_22 Q3(b); 9701_s23_qp_22 Q2(d); 9701_s21_qp_23 Q1(c); +10 additional hits |
| 27% |
12/44 |
14 |
major product / carbocation stability |
1. major product forms via the more stable carbocation/intermediate
2. more alkyl groups give greater positive inductive effect/electron donation
3. positive charge is spread/stabilised more |
9701_s20_qp_21 Q5(d)(ii); 9701_s20_qp_21 Q5(d)(i); 9701_m20_qp_22 Q3(c)(iii); +30 additional hits |
| 27% |
12/44 |
7 |
Kp / Kc expression and units |
1. for aA + bB ⇌ cC + dD, write Kc = [C]c[D]d / ([A]a[B]b)
2. for gases, write Kp = p(C)cp(D)d / (p(A)ap(B)b)
3. products over reactants; powers are stoichiometric coefficients
4. omit pure solids/liquids; derive units from the final expression |
9701_s23_qp_23 Q4(b)(ii); 9701_s21_qp_23 Q2(c)(iii); 9701_m20_qp_22 Q1(f)(ii); +9 additional hits |
| 25% |
11/44 |
12 |
SO₂ acid rain |
1. SO₂ reacts with water to form H₂SO₃
2. SO₂ / H₂SO₃ can be oxidised to H₂SO₄
3. these acids cause acid rain |
9701_w21_qp_23 Q1(d)(ii); 9701_w21_qp_21 Q1(d)(ii); 9701_s20_qp_22 Q2(d); +3 additional hits |
| 25% |
11/44 |
6 |
oxidising agent |
1. accepts electrons and is reduced |
9701_m21_qp_22 Q4(c)(i); 9701_m20_qp_22 Q2(c)(ii); 9701_m20_qp_22 Q2(b)(i); +11 additional hits |
| 25% |
11/44 |
7 |
Bronsted acid / strong vs weak acid |
1. acid = proton/H⁺ donor
2. strong acid fully dissociates/fully ionises in aqueous solution/water
3. weak acid partially dissociates/partially ionises in aqueous solution/water |
9701_w20_qp_23 Q2(b)(i); 9701_w20_qp_22 Q3(d)(ii); 9701_w20_qp_21 Q2(b)(i); +11 additional hits |
| 23% |
10/44 |
14 |
alkene test |
1. bromine water / Br₂(aq) is decolourised from orange/brown to colourless |
9701_s22_qp_22 Q6(a); 9701_s21_qp_21 Q5(c)(ii); 9701_s21_qp_21 Q4(b); +7 additional hits |
| 23% |
10/44 |
17 |
2,4-DNPH test |
1. 2,4-DNPH gives a red/orange/yellow precipitate with carbonyl compounds (aldehydes/ketones) |
9701_s21_qp_23 Q4(b); 9701_w20_qp_22 Q4(c); 9701_s20_qp_21 Q6(d); +10 additional hits |
| 23% |
10/44 |
19 |
nitrile hydrolysis to carboxylic acid / carboxylate |
1. acid hydrolysis: RCN + 2H₂O + HCl/H⁺ → RCOOH + NH₄Cl/NH₄⁺
2. alkaline hydrolysis: RCN + 2H₂O + NaOH → RCO₂Na + NH₄OH/NH₃; acidification gives RCOOH if required
3. heat/reflux with dilute acid or aqueous alkali
4. the nitrile carbon becomes the carboxyl carbon, so the carbon chain length does not change during hydrolysis |
9701_s25_qp_24 Q4(d)(i); 9701_s22_qp_22 Q5(c)(ii); 9701_w20_qp_21 Q3(c)(iv); +7 additional hits |
| 20% |
9/44 |
8 |
frequency of effective collisions |
1. rate increases/decreases with the frequency of successful/effective collisions per unit time |
9701_s22_qp_21 Q1(g); 9701_m22_qp_22 Q2(b)(iii); 9701_m21_qp_22 Q1(c)(ii); +7 additional hits |
| 20% |
9/44 |
11 |
NOₓ / atmospheric pollution |
1. NOₓ forms when N₂ and O₂ react at high temperature in an internal combustion engine / lightning
2. NO/NO₂ reacts with oxygen and/or water/moist air to form HNO₂/HNO₃ and cause acid rain
3. NO/NO₂ can act as a homogeneous catalyst in SO₂ oxidation to SO₃ and is regenerated
4. NO₂/NOₓ reacts with unburned hydrocarbons/VOCs to form photochemical smog / PAN |
9701_s24_qp_23 Q2(d)(i-ii); 9701_s23_qp_21 Q5(b)(i-iii); 9701_s22_qp_21 Q2(b)(i-ii); 9701_s20_qp_22 Q2(d); +5 additional hits |
| 20% |
9/44 |
9 |
Period 3 chloride hydrolysis |
1. SiCl₄ + 2H₂O → SiO₂ + 4HCl
2. PCl₅ + 4H₂O → H₃PO₄ + 5HCl
3. include state symbols / acidic products if the question asks |
9701_w21_qp_23 Q3(b)(ii); 9701_w21_qp_21 Q3(b)(ii); 9701_w20_qp_22 Q3(b)(ii); +12 additional hits |
| 20% |
9/44 |
17 |
Tollens / Fehling distinction |
1. aldehydes reduce Tollens’ reagent to a silver mirror/grey precipitate
2. aldehydes reduce Fehling’s solution to a brick-red precipitate
3. ketones show no visible change |
9701_m22_qp_22 Q4(a)(i); 9701_m21_qp_22 Q4(b); 9701_w20_qp_22 Q4(a)(iv); +6 additional hits |
| 20% |
9/44 |
2 |
relative atomic/isotopic/molecular mass |
1. mass / weighted average mass of atom, isotope or molecule
2. compared with 1/12 of the mass of a carbon-12 atom or the unified atomic mass unit |
9701_w22_qp_21 Q1(a)(ii); 9701_s20_qp_23 Q2(a); 9701_s20_qp_22 Q1(a); +6 additional hits |
| 18% |
8/44 |
14 |
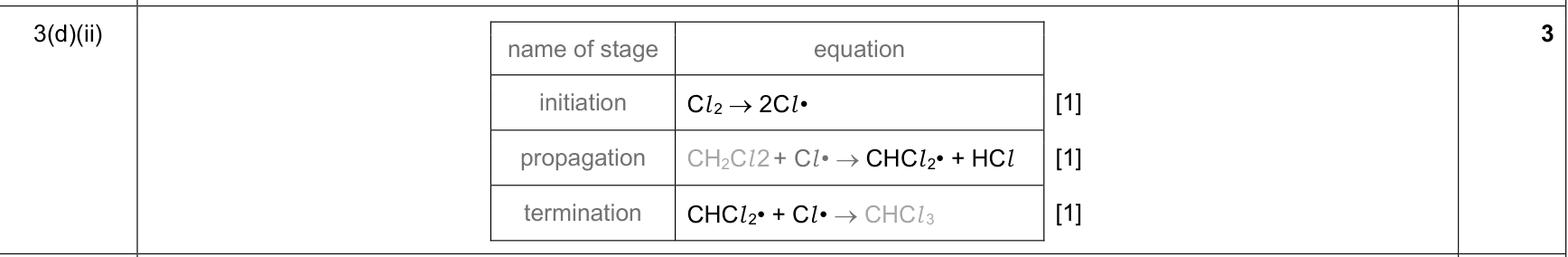
free-radical substitution |
1. initiation: homolytic fission in UV light forms halogen radicals
2. propagation 1: a halogen radical removes H from an alkane to form an alkyl radical and HCl/HBr
3. propagation 2: the alkyl radical reacts with Cl2/Br2 to form a halogenoalkane and regenerate a halogen radical
4. termination: two radicals combine to form a stable molecule |
9701_w24_qp_21 Q2(b)(iii); 9701_m21_qp_22 Q2(g)(i); 9701_s20_qp_23 Q4(b)(ii); +22 additional hits |
| 18% |
8/44 |
13 |
structural isomerism |
1. same molecular formula but different structural formulae |
9701_w22_qp_22 Q3(a)(ii); 9701_w22_qp_21 Q3(d)(iv); 9701_s21_qp_23 Q5(d); +6 additional hits |
| 18% |
8/44 |
4 |
ideal gas equation calculation |
1. use pV = nRT with Pa, m³ and K |
9701_m21_qp_22 Q1(b)(ii); 9701_m21_qp_22 Q1(b)(i); 9701_w20_qp_22 Q3(d)(iii); +3 additional hits |
| 18% |
8/44 |
5 |
enthalpy change of formation |
1. enthalpy/energy change when one mole of compound/substance is formed
2. from its elements in their standard states |
9701_w21_qp_22 Q1(b); 9701_s20_qp_21 Q3(a); 9701_m20_qp_22 Q1(g)(ii); +6 additional hits |
| 18% |
8/44 |
8 |
temperature effect |
1. higher temperature means particles have greater average kinetic energy, so a greater proportion of particles have E ≥ Ea
2. frequency of successful/effective collisions increases |
9701_s25_qp_21 Q3(a)(ii); 9701_s24_qp_23 Q2(b)(ii) |
| 16% |
7/44 |
12 |
photochemical smog / PAN |
1. unburned/unburnt hydrocarbons / VOCs react or combine with NO and/or NO₂
2. PAN / peroxyacetyl nitrate / peroxyacetylnitrate is formed
3. PAN is a component of photochemical smog |
9701_s25_qp_23 Q3(a)(ii); 9701_s24_qp_23 Q2(d)(ii); 9701_s22_qp_21 Q2(b)(i); 9701_m22_qp_22 Q2(d)(ii); +3 additional hits |
| 16% |
7/44 |
13 |
stereoisomerism |
1. same structural formula but different spatial arrangement of atoms/groups |
9701_w22_qp_23 Q5(a)(iii); 9701_w22_qp_21 Q5(a)(iii); 9701_w21_qp_22 Q3(e)(iv); +8 additional hits |
| 16% |
7/44 |
3 |
coordinate/dative bond definition |
1. a covalent bond/shared pair is formed
2. both electrons in the shared pair are donated by one atom/species |
9701_s21_qp_21 Q2(a)(i); 9701_m21_qp_22 Q2(e)(i); 9701_s20_qp_21 Q1(b)(ii); +4 additional hits |
| 14% |
6/44 |
11 |
AgNO₃ / NH₃ test |
1. acidify with dilute HNO₃, then add aqueous AgNO₃
2. Cl⁻ gives a white precipitate, soluble in dilute NH₃
3. Br⁻ gives a cream/off-white precipitate, partly/slightly soluble in concentrated NH₃
4. I⁻ gives a yellow precipitate, insoluble in concentrated NH₃ |
9701_w22_qp_21 Q2(c); 9701_s22_qp_22 Q2(d); 9701_m20_qp_22 Q2(c)(i); +6 additional hits |